Research Article
Yam Die-back and its Principal Cause in the Yam Belt of Nigeria
Plant Health Management Division, International Institute of Tropical Agriculture (IITA), PMB 5320, Ibadan, Nigeria
Yam die-back, is often referred to by a number of names including - yam necrosis, yam scorch, yam chlorosis and yam anthracnose. It is of wide distribution in the tropical regions of the world where yam (Dioscorea species) is grown. Yam die-back is a very destructive disease of D. alata (water yam) especially in the Carribeaps and the yam zone of tropical Africa (Fournet et al., 1974; Nwankiti and Arene, 1978). It was first reported on water yam in Nigeria in 1980 (Nwankiti and Okpala, 1981). It also attacks other yam species to a much lesser extent (Mignucci et al., 1988). Yield losses from the disease of up to 80 percent have been reported on different cultivars of D. alata in West Africa and Central America (Degras et al., 1984; Mignucci et al., 1988).
The disease can attack all parts of the yam plant at all stages of development. Infections concentrated or expressed on the shoot tips generally result in vine and/or tip die-back. Other symptoms expressed, generally depend on the age of the yam plant, the environmental conditions and the species and/or cultivar grown. Typically, when infection is initiated on the leaves, leaf symptoms first appear on lower older leaves close to the soil surface as pinpoint lesions that are dark brown, with or without pale brown or yellow chlorotic halos. These lesions expand rapidly and may coalesce, encompassing the entire leaf and resulting in premature leaf abscission. Severe infection results in defoliation leaving naked, black and drying vines. Shoot tip and stem die-back frequently occur under such severe infections. In some cases, there is a superficial bronzing and blackening of mature leaves on surfaces exposed to the sun, a condition often referred to as scorching. The disease reduces the effective photosynthetic surface of the plant and is particularly serious when it attacks the plant immediately after tuber initiation or during bulking (Hahn et al., 1987).
There has been some conflicting reports on the actual cause of yam die-back. Several have attributed the disease to be caused by a complex of organisms, the principal one being Colletotrichum species and thus have called it yam anthracnose (Jackson and Newhoot, 1978; Degras et al., 1984). Others have referred to it by the common names of yam chlorosis or necrosis (IITA, 1985) or yam scorch (Hahn et al., 1987; IITA, 1975), in reference to the symptoms observed on the leaves of infected plants. Others have even classified it as a virus disease caused by a potyvirus of unknown etiology (Theberge, 1985).
This confusion in the nomenclature of the disease due to uncertainties of its actual cause, necessitated an investigation into micro-organisms associated with naturally infected yam tissues exhibiting the typical symptoms associated with the disease. Pathogenicity tests were also carried out with sample isolates to establish with some certainty the main cause of the disease.
An extensive survey to determine the distribution of yam foliar diseases was made on farmers' fields across three ecological zones of Nigeria from August 19 to September 24, 1992 (Akem and Asiedu, 1993). During the survey, several samples of yam leaves and vines exhibiting varying symptoms of yam die-back were randomly collected from different locations and yam species. Isolations were made from the diseased plant samples collected, after incubation for 48 hrs in petri plates lined with moist filter paper. Sample pieces with spots or lesions were sterilely transferred to potato dextrose agar (PDA) after placing in 5 percent sodium hypochlorite for 5 minutes. The medium plates were incubated at 25±2°C for 5-7 days to enable the associated micro-organisms to grow out. Cultures obtained were further separated and purified into individual isolates. Purd cultures produced were identified after allowing to sporulate under continuous light at 27°C for 710 days. Subbultures were transferred to other PDA plates and placed in h refrigerator at 4 - 5°C for later use.
To prepare inoculum for spray inoculations, small samples from the refrigerated subcultures were transferred to PDA plates and left to sporulate under continuous light for 7-10 days at 27°C. lnoculum suspensions from the isolates were prepared by gently brushing off the surface of each culture plate with 50 ml of sterile distilled water using a camel's hair brush. The mycelia - spore suspensions were filtered through 2 layers of cheese cloth to remove mycelia. The resulting spore suspension of each microorganism was adjusted to 1.6 x 106 spores/ml using a haemacytometer. To determine the pathogenicity of the isolates obtained, Colletotrichum gloeosporoides, which was most prevalent among the isolates identified and its combination with other micro-organisms was, used to spray inoculate six cultivars of D. alata. These cultivars had been selected based on their observed reactions to yam die-back disease under natural field conditions. They were: Dan 087, TDa 86/00620 and TDa 316 observed to be resistant and TDa 85/00257, TDa 86/00607 and TDa 85/00250 observed to be susceptible to the disease (Akem and Asiedu, 1994). Minisetts were prepared from clean mature tubers of these cultivars using the minisett production procedure (Otoo et al., 1987). Following sprouting, four sets of each cultivar were transferred to steam - pasteurized soil in four plastic ,pots which were placed on the floor in a glass house. A small bamboo stake was used to support the sprouting set from each pot.
Cultures of C. gloeosporioides and other micro-organisms (Botryodiplodia theobromae, Fusarium oxysporum and Curvularia pallescens) identified from the samples, were used singly and in combination to spray-inoculuate the leaves and vines of the minisetts six weeks after transplanting to the pots. Thus the 5 treatment combinations administered were: C. gloeosporioides alone, C. gloeosporioides+B. theobromae, C. gloeosporioides+ F. oxysporium, C. gloeosporioides+B. theobromae, C. gloeosporioides+C. pallescens and C. gloeosporides+ B. theobromae , F. oxysporium+C. pallescens. Each plant was spray-inoculated with about 2 ml of spore suspension of each pathogen containing 1.6 x 106 spores/ml using a plastic 1L. hand-held sprayer. Unsprayed plants served as controls. All inoculated plants were first covered with polyethylene for 24 hrs before exposure to allow for symptom development. Pots were watered twice daily. Twenty eight days after spray inoculations, observable symptoms on all plants were recorded and the plants rated for disease reactions. A subjective scale of 0 to 5 was used as follows: 0.0 = no disease, no spots on leaves; 1.0 = slight disease, trace to 20 percent of leaves showing symptoms; 2.0 = moderate disease, 21-40 percent of leaves showing symptoms; 3.0 = severe disease, 41-60 percent of leaves showing symptoms with blackening of vines and some tip dieback; 4.0 = very severe disease, 61-80 percent of leaves showing symptoms with heavy vine and tip die-back; 5.0 = blighted conditions with 81-100 percent of leaves defoliated and whole plants dying or already dead with naked and black drying vines. Ratings were made to the tenth unit for close approximation of observable differences. Disease severity data were subjected to analysis of variance to determine least significant differences.
Diseased yam samples were collected during the survey from the 3 predominant yam species grown in Nigeria: D. alata, D. rotundata and D. dumetorum. Symptoms observed on these species varied greatly and seemed to be influenced by the ecology on which the yam species were grown. These variations were very striking on D. alata. In the southern forest region, symptoms on D. alata appeared mostly as streak browning lesions starting off on leaf veins and rapidly expanding to cover the entire leaf. Some local yam cultivars, especially "Ominelo" exhibited black coatings on the upper leaf surfaces exposed to the sun, qualifying for the common name of "yam scorch".
| Table 1: | Frequency of microorganisms isolated from diseased yam leaves and vines collected from farmers' fields in Nigeria |
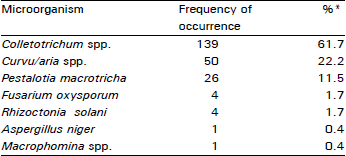 | |
| *Determined from a total of 225 culture isolates | |
| Table 2: | Frequency of different Colletotrichum species among Colletotrichum isolates from diseased yam leaf samples collected from farmers' fields |
 | |
| *Determined from 139 Colletotrichum species isolates | |
Generally, most cultivars, especially in the Southern Guinea savannas, where most of the yams are grown in Nigeria, exhibited the typical characteristic symptoms that start with circular black spots on leaf surfaces, expanding to leaf edge necrosis and then progressing to vine blackening and then tip die-back.
| Table 3: | Reaction of yam clones to micro-organisms isolated from diseased yam leaves and vines from farmers' fields |
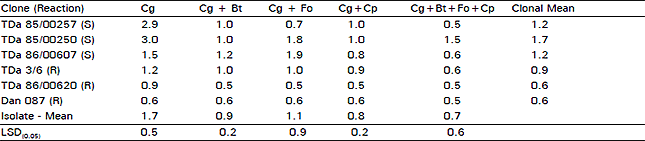 | |
S = Susceptible; R = Resistant; Cg = Colletotrichum gloeosporioides; Bt = Botryodiplodia theobromae; Fo = Fusarium oxysporum; Cp = Curvularia pallescens | |
On D. rotundata, typical symptoms observed were black circular spots, randomly distributed on leaf surfaces with extensive defoliation and vine blackening from severe infections. Some leaf scorching was also occasionally observed on some of the local clones. On D. dumetorum, circular spots with ash-gray centers were observed on infected leaves. Where attacks were severe, the leaves turned brownish-yellow followed by extensive defoliation.
A variety of micro-organisms were isolated from infected leaf and vine samples (Table 1). The frequency of occurrence of the different pathogens varied from less than 1 percent of the total samples to over 61 percent. The least frequently isolated micro-organisms were Aspergillus niger and Macrophomina spp, while the most frequently isolated were the Colletotrichum species (Table 1).
Four different species of Colletotrichum were identified from diseased yam leaf and vine samples collected. Of these four, C. gloeosporioides was the most frequent with a frequency of 84 percent while C. graminicola was the least with a frequency of about 3 percent (Table 2). C. gloeosporioides was also frequently isolated from the blackish surfaces of leaves often referred to as yam scorching. Colletotrichum species generally accounted for more than 61 percent of all the microorganisms isolated from the infected samples. When complex infections in which Colletotrichum was one of the component microorganisms were considered, this frequency was even higher.
Following spray-inoculations in the greenhouse, symptoms were expressed on infected plants within 10-14 days. Only the typical leaf necrosis and vine blackening symptoms were common on infected plants. Lesions began as specks with whitish centers surrounded by reddish margins and rapidly expanded to form patches on the more susceptible clones. Vine blackening followed severe infections but the typical tip die-back symptoms were not common. On the more resistant clones, initial specks did not expand any further. Under these controlled conditions, symptoms were first observed on young leaves, unlike in field natural conditions where infections are usually first observed on older lower leaves. The highest disease severity values were recorded on susceptible yam clones inoculated with C. gloeosporioides alone (Table 3). Reactions of these susceptible clones were generally weaker with pathogen combinations. The artificial inoculations confirmed reactions observed under natural field conditions (Akem and Asiedu, 1994) as clone ranking remained the same (Table 3). There were clearly significant differences (P<0.05) between susceptible and resistant clones inoculated with C. gloeosporioides. There was an apparent masking of the effects of C. gloeosporioides when it was combined with other micro-organisms isolated from same diseased samples. Clear differences between susceptible and resistant clones could not be detected with reactions from isolate combinations. Differences were only apparent when clonal disease severity means were considered.
From the symptoms observed on field infections and the high frequency of isolation of Colletotrichum species from the samples, Colletotrichum seem to be playing a major role in die-back disease of yam. This was also confirmed by artificial inoculations using isolates from the samples. Contrary to earlier reports. that there was no response to fungicide treatments (IITA, 1984), leading to the misleading conclusions that the disease was probably caused by a virus (Theberge, 1985), effective control of the disease has been obtained on D. alata diseased plots, with periodical sprays of the fungicide benlate. The erronuous concept that the disease was some kind of a complex of unknown etiology involving a virus has been quite costly to the yam breeding program at IITA. Valuable germplasm in the crossing blocks have been lost due to heavy attack from the disease which could have been protected with benlate sprays.
Colletotrichum species in general and C. gloeosporioides in particular, is responsible for most of the observed symptoms on yam leaves and vines that have led to the various names. All these names - yam necrosis, yam scorch, yam chlorosis and even the most common-yam die-back, are referring simply to yam anthracnose. If yam scientists would simply adopt and stick to the common name - yam anthracnose, for this disease, further confusion in the literature would be avoided.