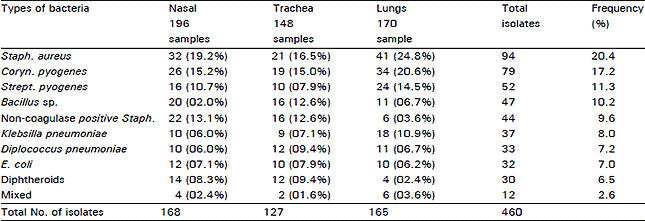
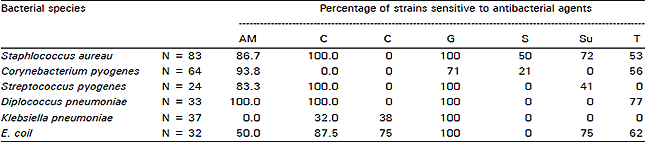
Research Article
Aetiological Study on Pneumonia in Camel (Camelus dromedarius) and in vitro Antibacterial Sensitivity Pattern of the Isolates
Department of Microbiology and Parasitology, College of Veterinary Medicine and Animal Resources King Faisal University, P.0. BOX 1757, Al-Asha 31982, Saudi Arabia
K. M. Mustafa
Department of Medicine, Pharmacology and Toxicology, Faculty of Veterinary Science, University of Khartoum, Sudan
G. E. Mohamed
Department of Medicine, Pharmacology and Toxicology, Faculty of Veterinary Science, University of Khartoum, Sudan