Research Article
B-carotene Lock the Effect of Acrylamide on Liver in the Egyptian Toad
Department of Zoology, Faculty of Science, Alexandria University
A. Abou-Gabal
Department of Genetics, Faculty of Agriculture, Tanta University, Egypt
Environmental compounds are known to be involved in the generation of many cancers. Acrylamide is widely used in the synthesis of variety of polymers, some of which are used as coagulant aids in the treatment of drinking water (McCollister et al., 1965). Little attention has been given to the carcinogenicity of acrylamide (Bull et al., 1984a).
Elimination of carcinogenic compounds from our environment would be expected to help prevent human cancer, but this is not a practical proposition. Therefore, it is important to discover naturally occurring or synthetic compounds which can suppress or prevent the process of carcinogenesis (Hill and Grubbs, 1992). Epidemiological studies have shown that intake of retinoid as well as their precursor B-carotene reduce the risk of cancer (Bollag and Hartmann, 1983). B-carotene and other carotenoids have been found to posses common biological functions (Photoprotection, antioxidant properties, immunomodulation and anti-cancer activity) in both humans and rodents (Sadek, 1989; Lim et al., 1992).
On the other hand, some studies on the effect of retinoid on carcinogenesis have given apparently contradictory results. In tumor promotion studies, retinoid both inhibit (Hill and Grubbs, 1992) and enhance (Hasegawa et al., 1988) the induction of tumors in toads and human were reported (Abdelmeguid et al., 1997).
The present study therefore was undertaken to determine whether acrylamide have any carcinogenic effects on liver of the Egyptian toad, Bufo regularis and to clarify the role of B-carotene on the liver in the experimental toads, previously treated with acrylamide.
Sexually mature male and female, B. regularis, with 30 g average weight were collected by a regular supplier from AlNozha District, Alexandria, Egypt. The toads were maintained in glass tanks at temperature 20-22°C. Mud and stones were placed on the bottom of the aquaria to approximate the natural environment of the animal. Dead toads were quickly removed from the aquaria the toads were fed on earthworms once every 3 days. The experimental animals were divided into 5 groups (50 toads/group) and treated as follows:
| 1. | The animals of group A were injected into the dorsal lymph sacs with acrylamide (Sigma Chemical Company St. Louis, MO, USA) at a dose of 005 mg/toad, twice/week for 12 weeks |
| 2. | Toads of group B were given the same dose of acrylamide and injected with 0.05 mg B-carotene (BC) (Sigma Chemical Company, St. Louis, MO, USA), 3 hr, prior to the carcinogen, twice/week for 12 weeks |
| 3. | Animals of group C were given the same dose level of acrylamide and. BC 3 hr. after the carcinogen, twice/week for the same period |
| 4. | Animals of group D were treated with BC alone at the same dose and for the same period as the animals of group B or C |
| 5. | This group of animals (Group E) have not been treated with anything and used as control |
At the end of 12 weeks, all animals were killed. The organs were examined by the naked eye. In the liver of some animals only a single tumor appeared, while in others several tumors of different sizes appeared. These tumors were greyish white in colour. For histological evaluation, the liver was fixed in Bouin, embedded in paraffin and sectioned 4 pm thick, slides were stained with hematoxylin and counter-stained with eosin.
The present results showed that toads which injected with 0.05 mg acrylamide/toad, twice/week for 12 weeks, induced liver tumors in 9 cases out of 50 cases (Table 1). The liver tumors were diagnosed as hepatocellular carcinoma. On the other hand the carcinogenic effect of acrylamide was completely blocked in toads which injected with 0.05 mg B-carotene, 3 hr prior to the carcinogen, twice/week for 12 weeks. However, toads treated with acrylamide at the same dose level and 0.05 mg of BC, 3 hr.after the carcinogen, twice/week for 12 weeks showeda lower incidence of liver tumors.
Neither tumor growth nor neoplastic changes were detected after 12 weeks in the liver of toads which were given 0.05 mg of BC/toad, twice/week.
The results of the present study clearly indicate that acrylamide induced liver tumor in the Egyptian toad. The tumorigenic activity of acrylamide in skin and lung of SwissICR mice was studied, by Bull et al. (1984a). Also, the carcinogenicity of acrylamide in SENCR and A/J mice strain has been described before (Bull et al., 1984b).
| Table 1: | The role of B-carotene (BC) on toad liver tumor induced by acrylamide (AC) |
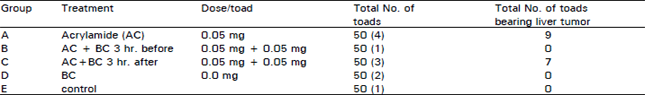 | |
| ( ): number of dead toad. *Not significant, as compared with the a crylamide - alone group | |
The present investigation demonstrated that B-carotene inhibited completely toad liver tumor when administered before the carcinogen (acrylamide) and that it was less effective when administered after the carcinogen. It has been shown that B-carotene have a predictive actions against carcinomas in the skin, colo, buccal pouch epithelia, mammary gland, salivary gland and liver in experimental animals (Hill and Grubbs, 1992).
On the other hand, Moon (1989) reported no effect of B-carotene on N-butyl-N (4-hydroxybutyl) nitrosamine (OHBBN)-induced bladder caTcinogenesis. Similarly, no discernible effects on the growth rate of Morris transplantable hepatoma could be observed following dietary administration of B-carotene for six weeks (Blakely et al., 1988).
The mechanism by which BC inhibits liver tumor in initiation, or post-initiation are unknown. There is increasing interest in the rate of antioxidant vitamins like ascorbic acid, atocopherol, retinal and BC in neutralizing free radicals and overtly aggressive oxygen species (Burton and Ingold 1984). Free radicals and non-radical oxidizing species are regularly produced in animals treated with carcinogens (Sato, 1989) and also in human tissues (Ames and Saul, 1987) which are capable of damaging DNA, protein, lipids and gene disposition. Furthermore, BC is among the most efficient substance known for quenching the excitation energy of single oxygen and also for trapping certain organic free radicals. It has a direct inhibitory effect on liver microsomal enzymes (Temple and Joyce, 1987), thus offering another mechanism of its anticancer nature.
It is concluded that the time of dosing with BC is often important. To be most effective in preventing toad liver tumor, BC should be applied shortly initiation. Much additional study is required to determine its effect on genetic disposition.