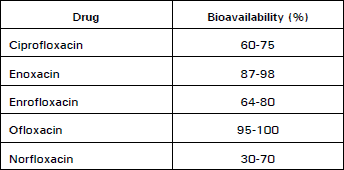
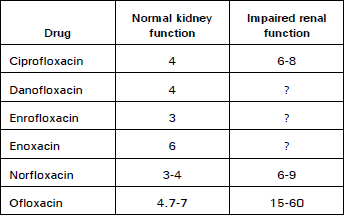
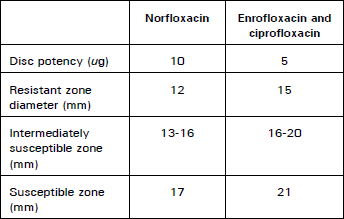
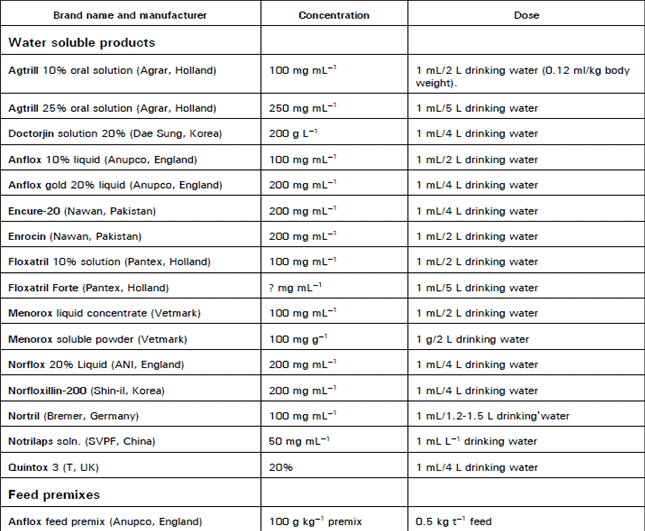
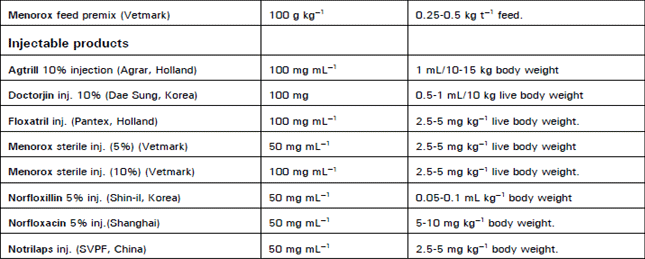
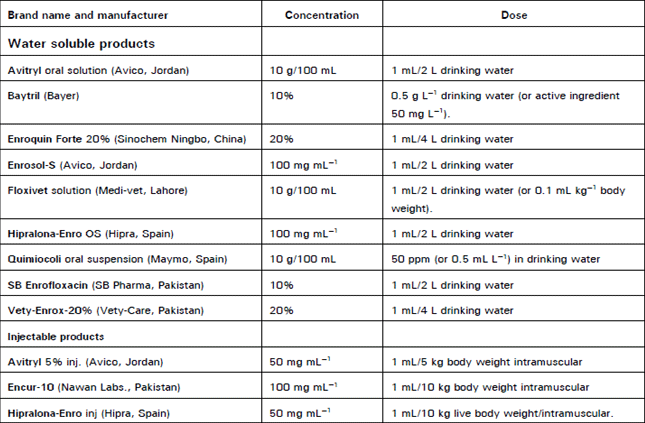
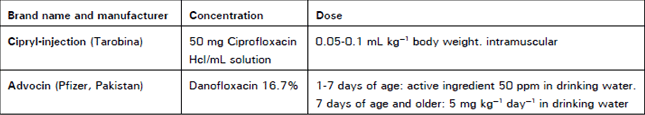
Clinical Report
Use of second generation quinolones in Poultry
Department of Veterinary Pathology, University of Agriculture, Faisalabad-38040, Pakistan
Farzana Rizvi
Department of Veterinary Pathology, University of Agriculture, Faisalabad-38040, Pakistan