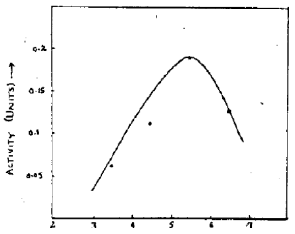
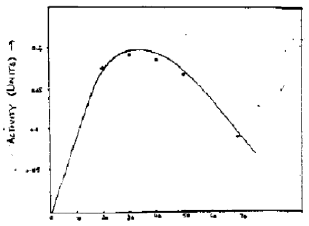
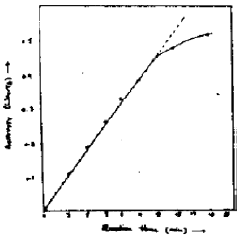
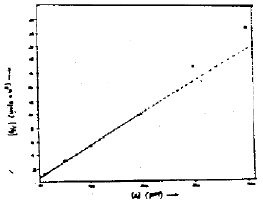
Research Article
Isolation and Characterization of Acid Phosphatase from Wheat (Pirsabak-85) Grains (Triticum aestivum)
Department of Chemistry, University of Peshawar, Pakistan
Razia Khan
Department of Chemistry, University of Peshawar, Pakistan
Mohammad Tariq
Department of Chemistry, University of Peshawar, Pakistan
S.F. Mabood
Department of Chemistry, University of Peshawar, Pakistan