Research Article
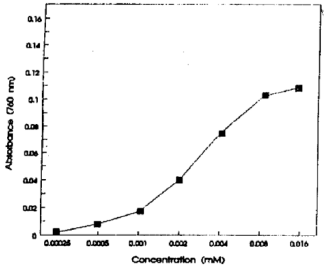
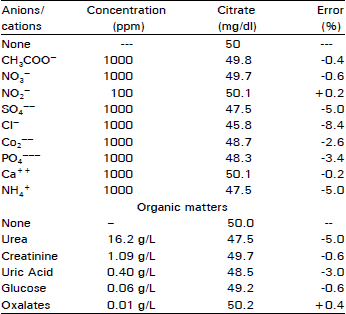
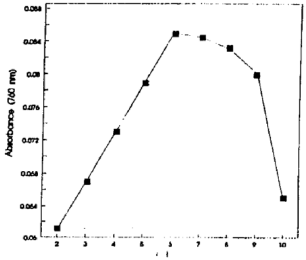
Studies on the Photometric Estimation of Citrate in Urine
Institute of Biochemistry,Department of Urology, Bolan Medical College, Quetta, Pakistan
Nabeel Y. Khalil
Institute of Biochemistry,Department of Urology, Bolan Medical College, Quetta, Pakistan
Sher Ahmed
Department of Urology, Bolan Medical College, Quetta, Pakistan
Fouzia S. Rehmani
Department of Urology, Bolan Medical College, Quetta, Pakistan
M. Yaqoob
Department of Chemistry, University of Balochistan, Quetta,Department of Urology, Bolan Medical College, Quetta, Pakistan
M. Masoom Yasinzai
Institute of Biochemistry, Department of Urology, Bolan Medical College, Quetta, Pakistan