Research Article
Effects of Salt on Seed Germination in Contrasting Cereal Cultivars
Government College Faisalabad-Pakistan
M. S. Davies
School of Pure and Applied Biology, University of Wales, U.K
Osmotic and specific effects are the most frequently suggested mechanisms by which saline substrates reduce plant growth. However, the relative importance of osmotic and specific ion effect on plant growth seems to very depending on the drought and/or salt tolerance of the plant under study. When seeds are moistened with a saline solution, their germination is affected by the high internal concentration of toxic ions and the low external water potential. Salt tolerance during the germination stage is critical for plant growing in saline soils. The heritability of this trait has been demonstrated in several species: for instance in rice Akbar (1985). Some varieties within a single species e.g. rice (Yeo et al., 1990), wheat (Rashid, 1986) and Sorghum (Yang et al., 1990) exhibit very different levels of tolerance to external NaCl. There has been a considerable research on salt tolerance in natural species, among ecotypes within species and in cultivars of crop plants. Information has also been gathered on the physiological and biochemical mechanisms of salt tolerance (Ashraf et al., 1986; Yang et al., 1990).
A number of characters have been used to screen plants for salt tolerance (Yeo et al., 1990). They have included modifications of a rooting test (Hannon and Bradshaw, 1968; Tiku and Sanaydon, 1971) and salt accumulation/exclusion (Greenway and Munns, 1980; Yeo et al., 1990) and yield response (Chauhan, 1987).
The aim of these experiments were to measure the salt tolerance of various species-cultivars at germination stage.
The seeds of Secale cereale cv. K2 (2n = 2x = 14) and wheat. (Triticum aestivum cv. Chinese spring), Triticum aestivum (2n = 6x = 42) cv. Lyallpur 73, Pak 81 and Lu-26-S and the seeds of barley (Hordeum sativum) (2n = 2x = 14) and the cultivar Sandal (2n = 6x = 42) of wheat were obtained from Plant Breeding international Cambridge U.K. Punjab Agriculture Research Institute, Faisalabad and from Soil Science Department, University of Agriculture Faisalabad, respectively. The seeds of uniform size and appearance were sown in different experiments on rafts (75 mm diameter) consisting of fibre glass tissue stretched across and glued with cow gum to a ring of expanded polystyrene. Rafts were floated on 1 dm3 of nutrient solution contained in plastic boxes (210× 140× 80 mm). The solution was continuously and gently aerated by bubbling air through diffusing stones using aquanum pumps.
In general there were six rafts in each plastic box and the number of seeds on each raft were 10. The varying NaCl concentrations were supplied in a background of 0.1 strength nutrient solution. The experiments were carried out in a growth chamber at a constant temperature of 20+0.5°C with illumination provided for 16 h per day by white fluorescent tubes. The period of exposure of NaCl was 8 days after sowing. Seedlings were harvested randomly per treatment per cultivars and were immediately fixed in 3:1 v/v ethanol:glacial acetic acid mixture and stored at 4°C.
Secale cereale (cv. K2) and Triticum aestivum
cv. Chinese Spring (0-200 mM NaCl)
The percentage of seeds germinating declined significantly by NaCl concentrations (Fig. 1). Germination of T. aestivum cv. Chinese Spring was 66.7 in the control (0 mM NaCl), this declined to 31.7 at 50 mM NaCl and a further decline to 8 occurred at 150 mM culminating in a germination of 3 at 200 mM. The pattern of reduction in seed germination with increasing NaCl concentration in S. cereale cv. K2 was essentially similar to that of T. aestivum cv. Chinese Spring. The NaCl term in the analysis was highly significant (p<0.001). Neither the species term nor the species × NaCl concentration term of the analysis of variance was significant (p>0.05).
The data showed that the NaCl concentration range used was too high to test the effect of NaCl on cellular process.
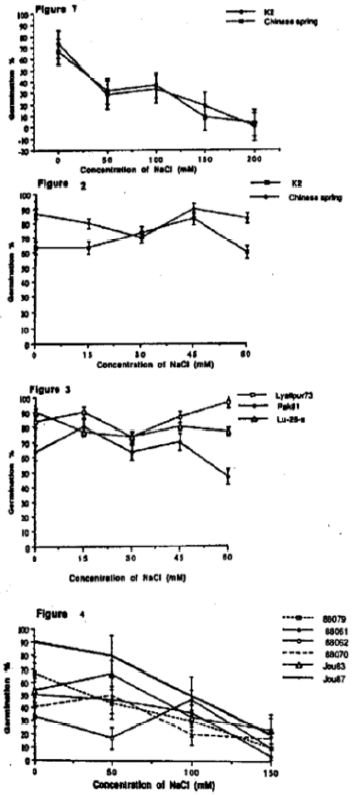 | |
| Fig. 1-4: | The cumulative percentage of seeds germinating of Secale cereale cv. k2, Triticum aestivum cvs, chinese spring, Lyallpur 73, Pak 81, Lu-26-s, and Hordeum sativum line. B-88061, B-88062, B-88079, B-jou 83 and B-jou 87 on experimental day 8, when grown in solutions containing 0-200 mM NaCl supplied in a background of 0.1 strength Rorison’s solution. The vertical bars represent ±SE. |
Consequently, a further experiment was carried out to determine the effect of a narrower range of salt concentration (0-60 mM NaCl) on germination.
Secale cereale (cv. K2) and Triticum aestivum (cv. Chines Spring) (0-60 mM NaCl):
The NaCl term in the analysis of variance was non significant. However, the species term was significant (p<0.05) but the species x NaCl concentration interaction was non significant.
The percentage of seeds germination, in both the species, was not generally affected by increasing NaCl concentration (Fig. 2). Generation in T. aestivum cv. Chinese Spring and S. cereale cv. K2 was 86.6 and 63.3% in the control solution (O NaCl) respectively. Triticum, had a greater percentage germination (86.7%) that of Secale (63.3%) in the control (O NaCl). Triticum aestivum cv. Chinese Spring also had a significantly higher percentage germination in the increasing NaCl concentration then that of Secale. The percentage of germination of T. aestivum cv. Chinese Spring was 80, 70, 90 and 83.3% of the control at 15, 30, 45 and 60 mM NaCl respectively, whereas the corresponding values of S. cereale cv. K2 were 63.3, 73.3, 83.3 and 60%.
Triticum aestivum (cvs. Lyallpur 73, Pak 81 and Lu-26-S) (0-60 Mm NaCl):
The NaCl term in the two analysis of variance was non significant although the cultivar term was significant (p<0.05). The cultivar x NaCl was non significant.
The percentage of seed germinating in three cultivars of Pakistani wheat, Lyallpur 73, Pak 81 and Lu-26-S, was not strongly influenced by increasing NaCl concentration (Fig. 3) although the germination of Lyallpur 73 increased slightly at 60 mM NaCl.
Triticum aestivum (cv. Sandal) (0-60 mM NaCl):
The percentage of seed germinating in T. Aestivum cv. Sandal was relatively unaffected by increasing NaCl concentration. The cumulative percentage germination of cultivar Sandal was 91.7, 86.7, 88.1, 75 and 83.2% at 0, 15, 30, 45 and 60 mM NaCl concentration term in one way analysis of variance was non significant.
Hordeum sativum (lines. B-88061, B-88062, B-88070, B-jou83) and B-jou87 (0-60mM NaCl):
The percentage of seed germination in the barley lines was not strongly affected by increasing NaCl concentration. The lines, B-jou87 and B-88079, showed a greater overall germination than the other lines tested. The NaCl term in the analysis was significant (p<0.05). However, although the lines terms in the AOV was highly significant (p<0.001), the line x NaCl concentration interaction was non significant.
As NaCl concentration over range 0-60 mM had little effect on seed germination in these barley cultivars, it was decided to examine their response over a wide rage of NaCl concentrations (0-150 mM).
Hordeum sativum (lines. B-88079, B-88061, B-88062, B-88070, B-jou83) and B-jou87 (0-150 mM NaCl): There was a highly significant (p<0.001) difference between lines but the interaction line vs NaCl was non significant.
The cumulative percentage germination in the barley lines was significantly reduced by increasing NaCl concentration at 150 mM NaCl (Fig. 4) in all of the lines. There were significant differences between the lines in the percentage germination in the control treatment and at 50 mM NaCl, for example, B-jou87 had 90% germination at 0 NaCl compared with 33.3% in B-88062.
Seed germination was relatively insensitive to increased NaCl over the range 0-60 mM NaCl is wheat, barley and rye, but concentration above 60 mM (60-150 mM) resulted in a significant reduction in the proportion of seeds germination. In general the lines of barley which were used were less sensitive to increased NaCl in respect of this character than wheat or rye. NaCl reduction in seed germination has also been reported by other workers: in Triticum (Kingsbury and Epstein 1986) and in barley (Bhatti et al., 1976). The basis for this reduced germination may be due to increased internal concentration of Na+ and Cl– and thereby toxicity of these ions to physiological process, but may also be influenced by osmotic effects due to higher concentration of ions in the surrounding solution , as well as osmotic effects caused by higher internal ion concentration. It is not possible in this and indeed in most other studies, of this phenomenon to distinguish critically between these effects. Although, germination was reduced by salinity, there was comparatively little difference between the cultivars within each species in their response to NaCl. Thus in these studies the proportion of seeds germinating was not a good discriminating character for assessing the salt tolerance of these crop cultivars.
M. Hanif thanks the Government of Pakistan for the award of Central Overseas Training Scheme Scholarship for Ph. D in U.K.