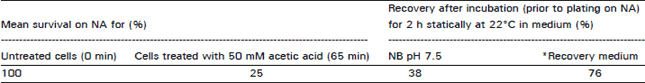
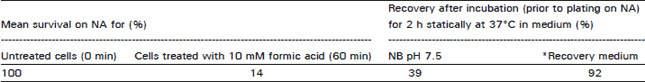
Research Article
Recovery of Injured Escherichia coli
ABI, Nat. Agric. Res. Centre, P.O NARC, Park Road, Islamabad, Pakistan
R.J. Rowbury
Biology Department (Darwin Building), University College London, Gower Street, London WC1E 6BT, U.K
D.G. Smith
Biology Department (Darwin Building), University College London, Gower Street, London WC1E 6BT, U.K