Research Article
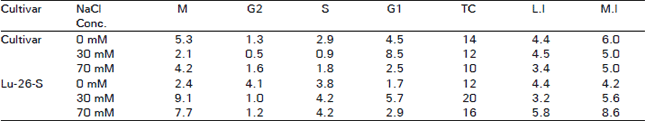
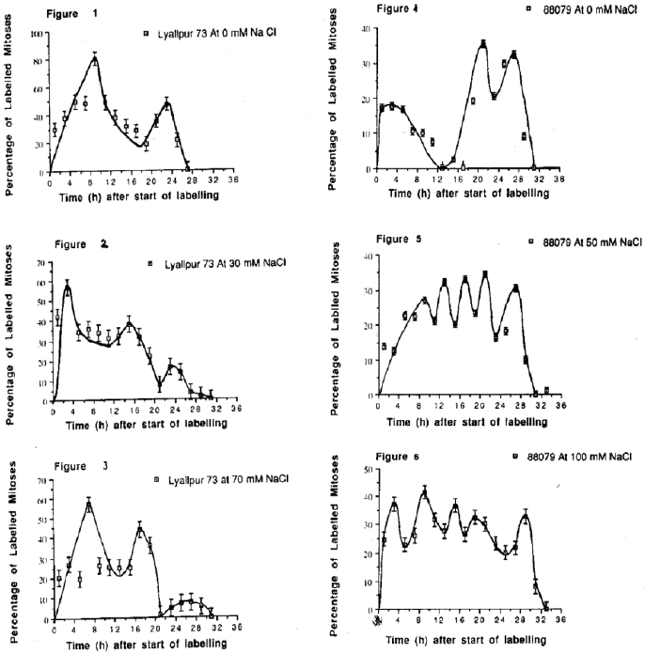
The Influence of NaCl on the Cell Cycle and Growth Fraction in the Root Meristems of Triticum aestivum cvs. Lyallpur 73 and Lu-26-S
School of Pure Applied Biology, University of Wales, Cardiff, UK.
M. S. Davies
School of Pure Applied Biology, University of Wales, Cardiff, UK.