Research Article
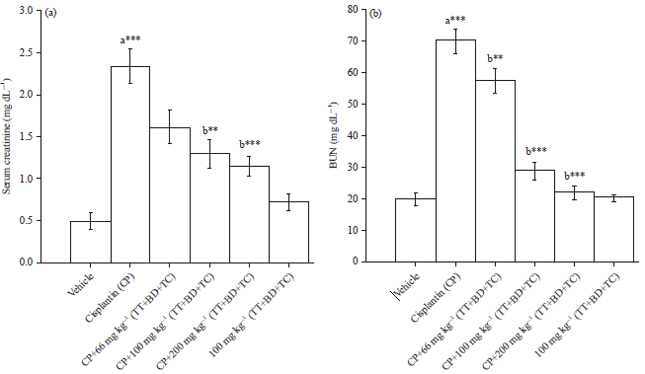
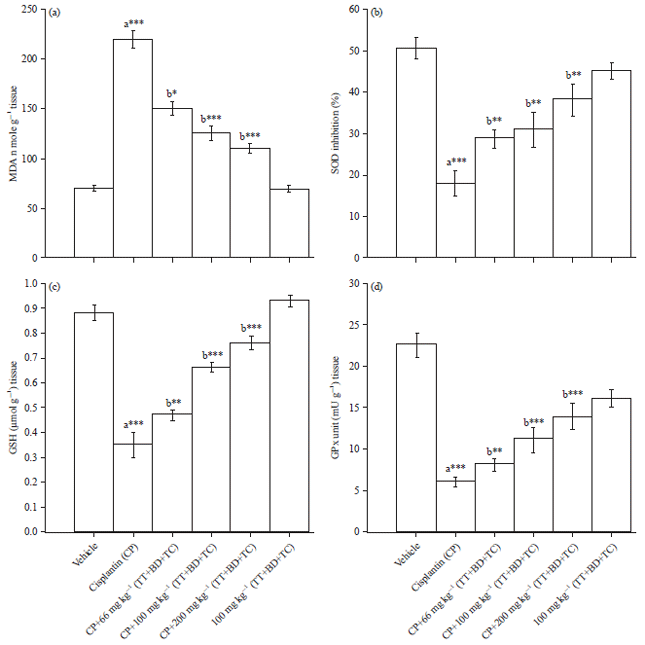
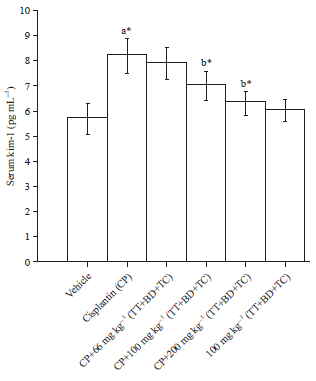
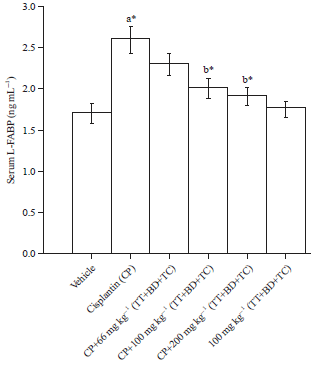
Combination of Tribulus terrestris, Boerhaavia diffusa and Terminalia chebula Reverses Cisplatin-induced Nephrotoxicity in Wistar Rats
Department of Pharmacology, All India Institute of Medical Sciences, New Delhi 110029, India
Harlokesh Narayan Yadav
Department of Pharmacology, All India Institute of Medical Sciences, New Delhi 110029, India
LiveDNA: 91.817
Vishwa Mohini Khare
Department of Radiology, School of Medicine, University of Pennsylvania, Philadelphia, PA 19104, USA
Surender Singh
Department of Pharmacology, All India Institute of Medical Sciences, New Delhi 110029, India
Yogendra Kumar Gupta
Department of Pharmacology, All India Institute of Medical Sciences, New Delhi 110029, India
LiveDNA: 91.10871
Amit K. Tiwari
Department of Pharmacology and Experimental Therapeutics, The University of Toledo, Toledo, OH 43614, USA
LiveDNA: 91.26934