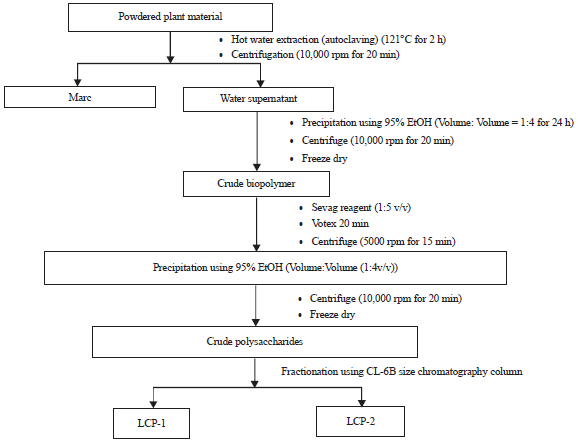
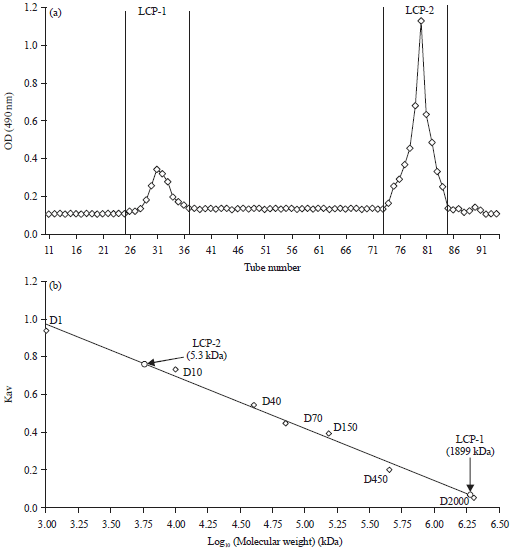
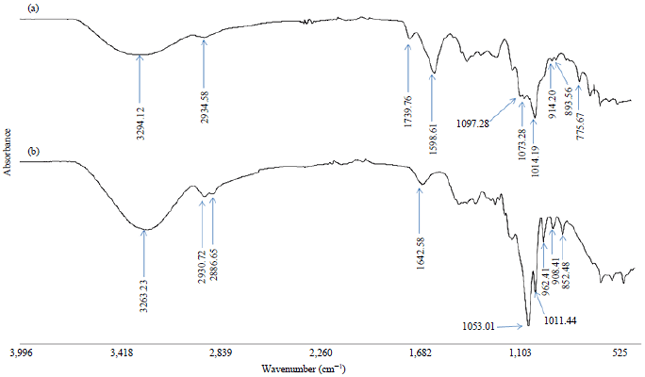
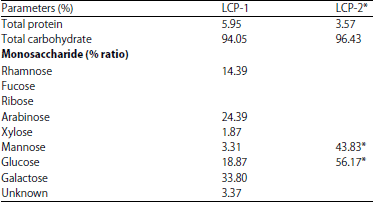
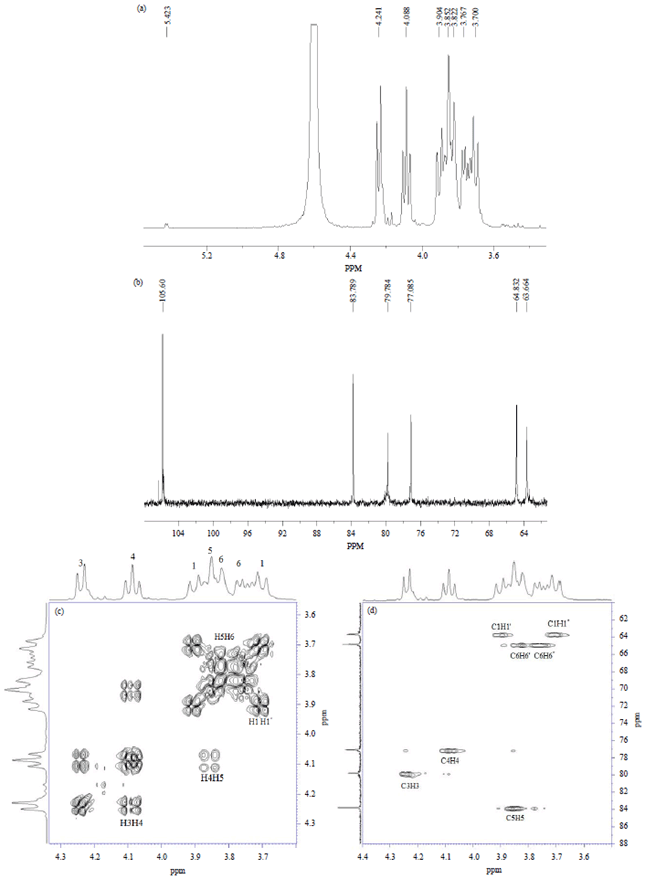
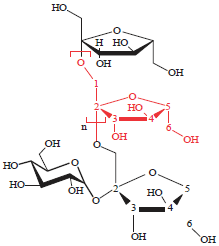
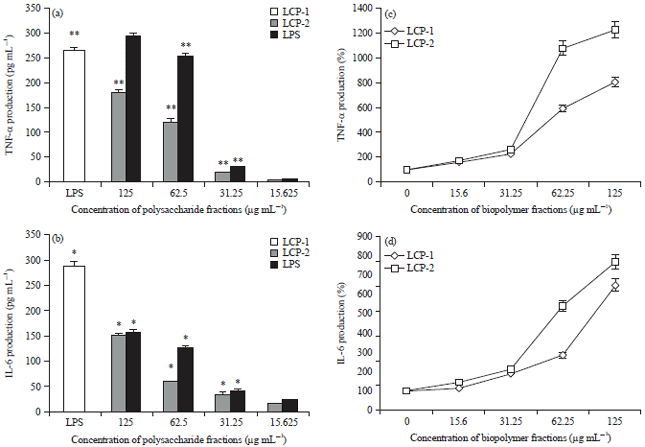
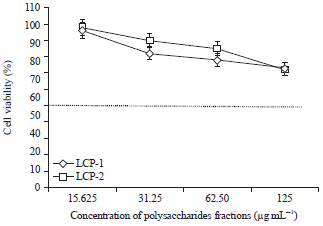
Research Article
Antioxidant and Immunomodulatory Activities and Structural Characterization of Polysaccharides Isolated from Lobelia chinensis Lour
School of Science and Health, Parramatta Campus, Western Sydney University, Locked Bag 1797, Penrith NSW 2751, Australia
Narsimha Reddy
School of Science and Health, Parramatta Campus, Western Sydney University, Locked Bag 1797, Penrith NSW 2751, Australia
LiveDNA: 91.20335
Cheang Soo Khoo
WentworthInstitute, 302-306 Elizabeth Street, Surry Hills, NSW 2010, Australia
Sundar Rao Koyyalamudi
Institute of Endocrinology and Diabetes, The Children�s Hospital at Westmead, Sydney, NSW 2145, Australia
LiveDNA: 61.6258
Christopher E. Jones
School of Science and Health, Parramatta Campus, Western Sydney University, Locked Bag 1797, Penrith NSW 2751, Australia