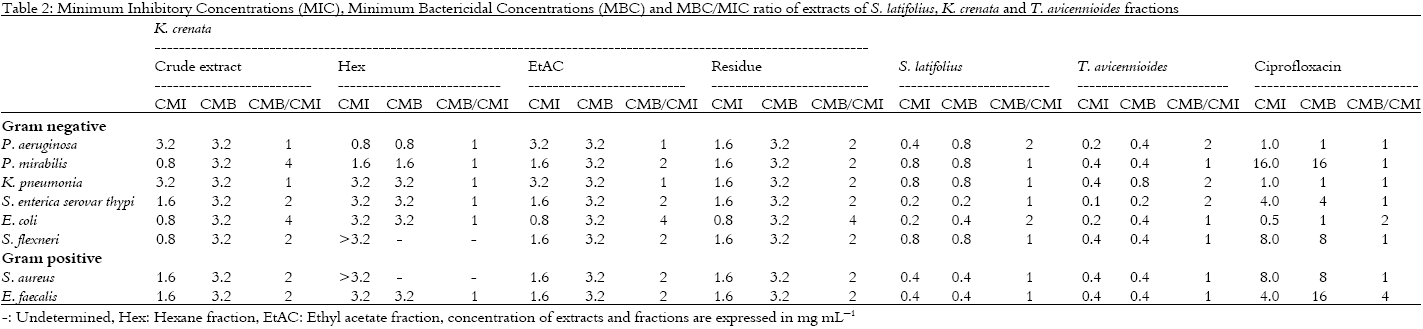
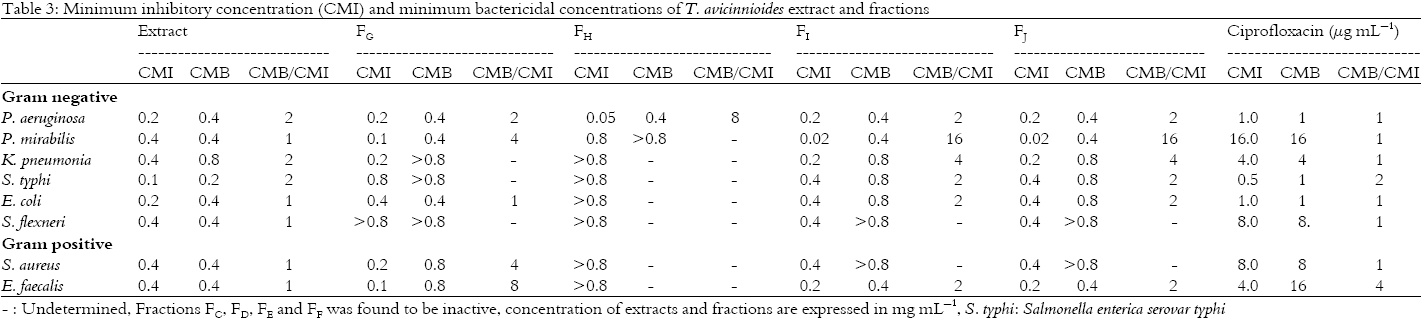
Research Article
Antibacterial Activity of Methanol Extracts and Fractions from Kalanchoe crenata, Terminalia avicennioides and Sarcocephalus latifolius
Laboratory of Pharmacognosy, University of Montains, P.O.Box 208, Bangangte, Cameroon
Mouokeu Raymond Simplice
Laboratory of Microbiology and Food Quality Control, Institute of Fisheries and Aquatic Sciences, University of Douala, P.O.Box 7236, Douala, Cameroon
Nguimatsia Francois
Laboratory of Pharmacognosy, University of Montains, P.O.Box 208, Bangangte, Cameroon
Njateng Guy Sedar Singor
Laboratory of Microbiology and Antimicrobial Substances, Faculty of Sciences, P.O.Box 67, Dschang, Cameroon
Tamokou Jean de Dieu
Laboratory of Microbiology and Antimicrobial Substances, Faculty of Sciences, P.O.Box 67, Dschang, Cameroon
Kuiate Jules Roger
Laboratory of Microbiology and Antimicrobial Substances, Faculty of Sciences, P.O.Box 67, Dschang, Cameroon