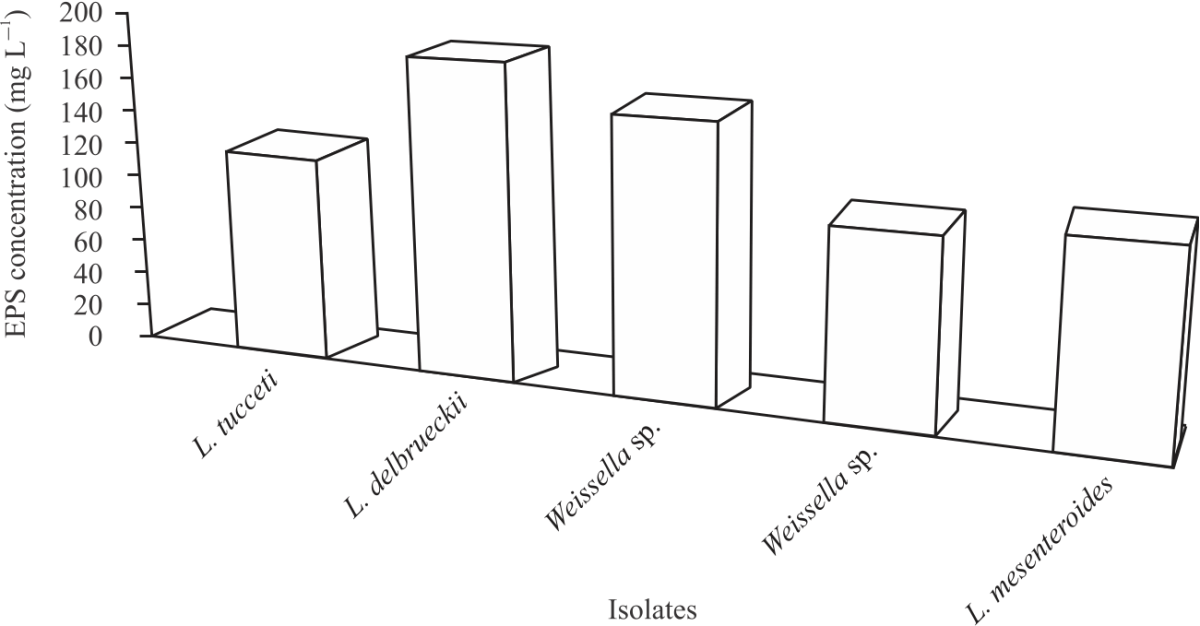
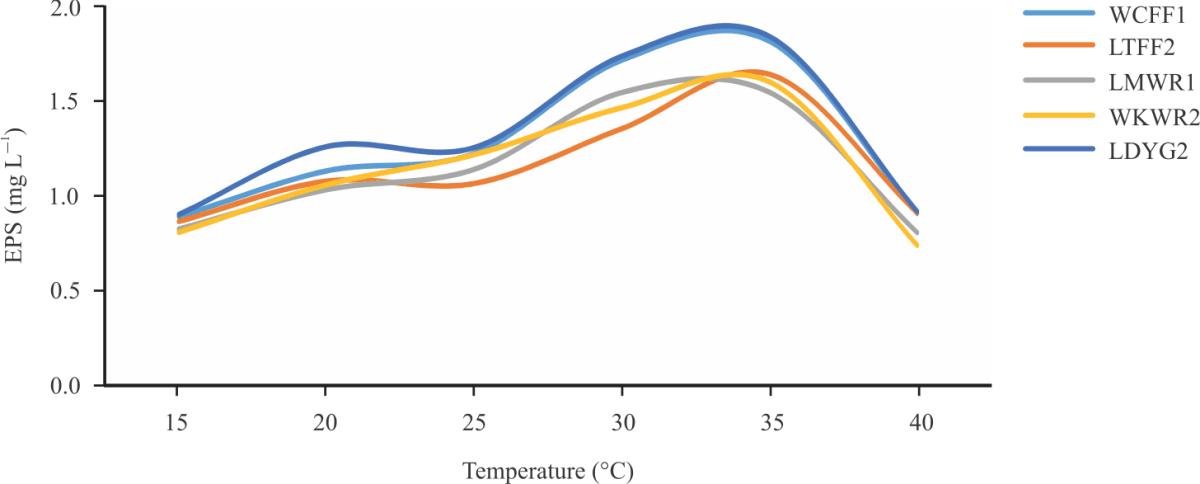
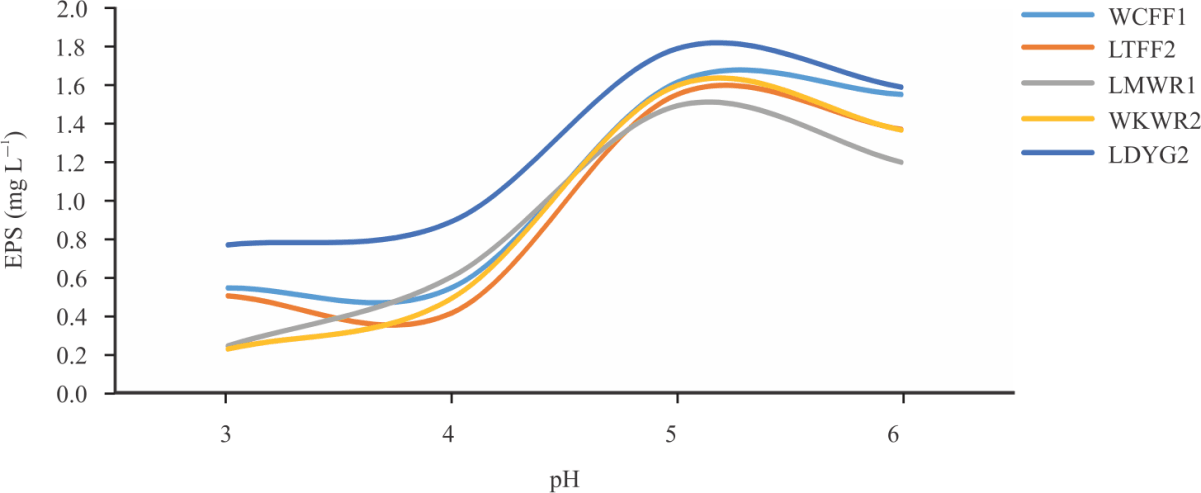
Research Article
Optimization of Extracellular Polysaccharide Substances from Lactic Acid Bacteria Isolated from Fermented Dairy Products
Department of Biological Sciences, Ajayi Crowther University, Oyo, Nigeria
LiveDNA: 234.34604
Bukola Adebayo-Tayo
Department of Microbiology, University of Ibadan, Oyo State, Nigeria
Jadesola Sanusi
Department of Biological Sciences, Crescent University, Abeokuta, Nigeria