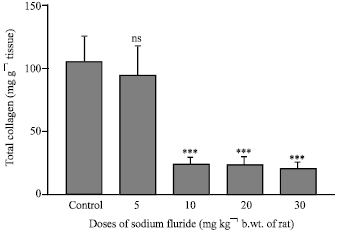
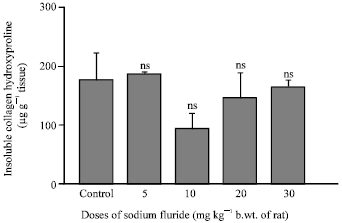
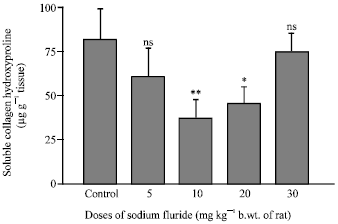
Research Article
Effect of Different Doses of Sodium Fluoride on Various Hydroxyproline Fractions in Rat Kidneys
Department of Biochemistry, College of Science, King Saud University, P.O. Box 2455, Riyadh-11451, Saudi Arabia
N.J. Siddiqi
Department of Biochemistry, College of Science, King Saud University, P.O. Box 2455, Riyadh-11451, Saudi Arabia
A.S. Alhomida
Department of Biochemistry, College of Science, King Saud University, P.O. Box 2455, Riyadh-11451, Saudi Arabia