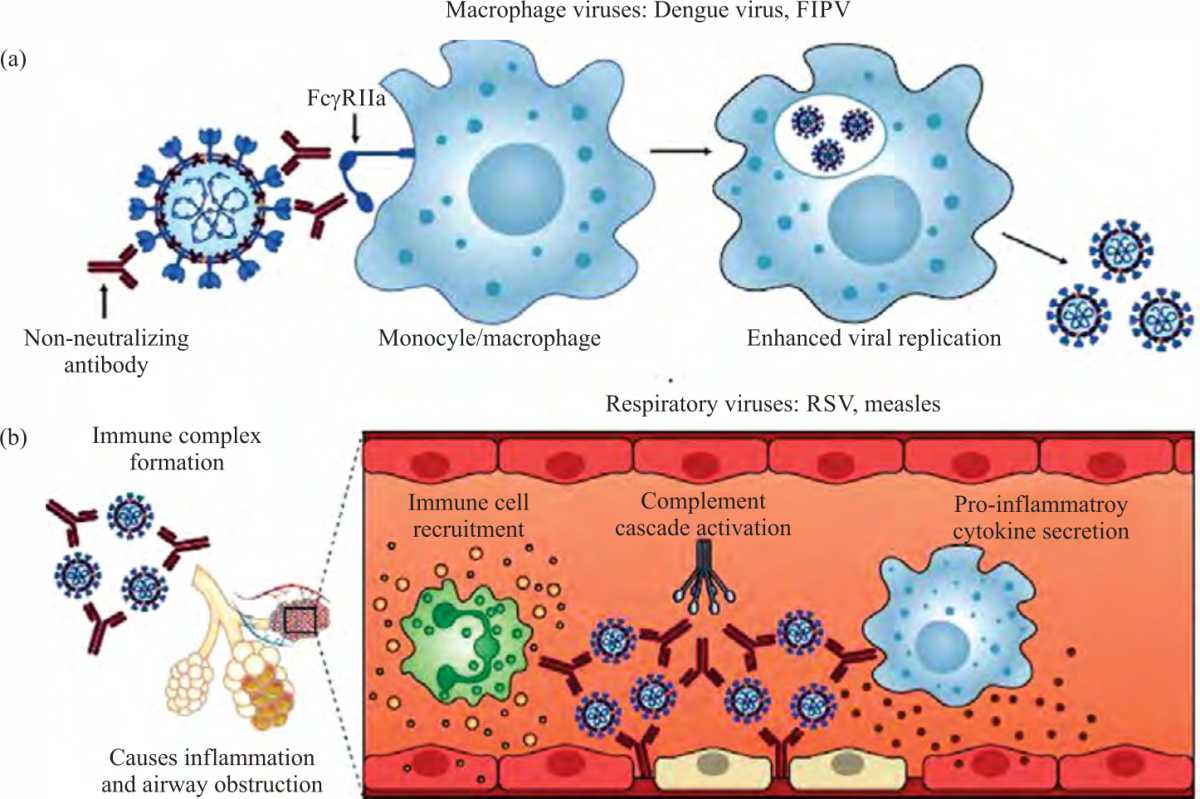
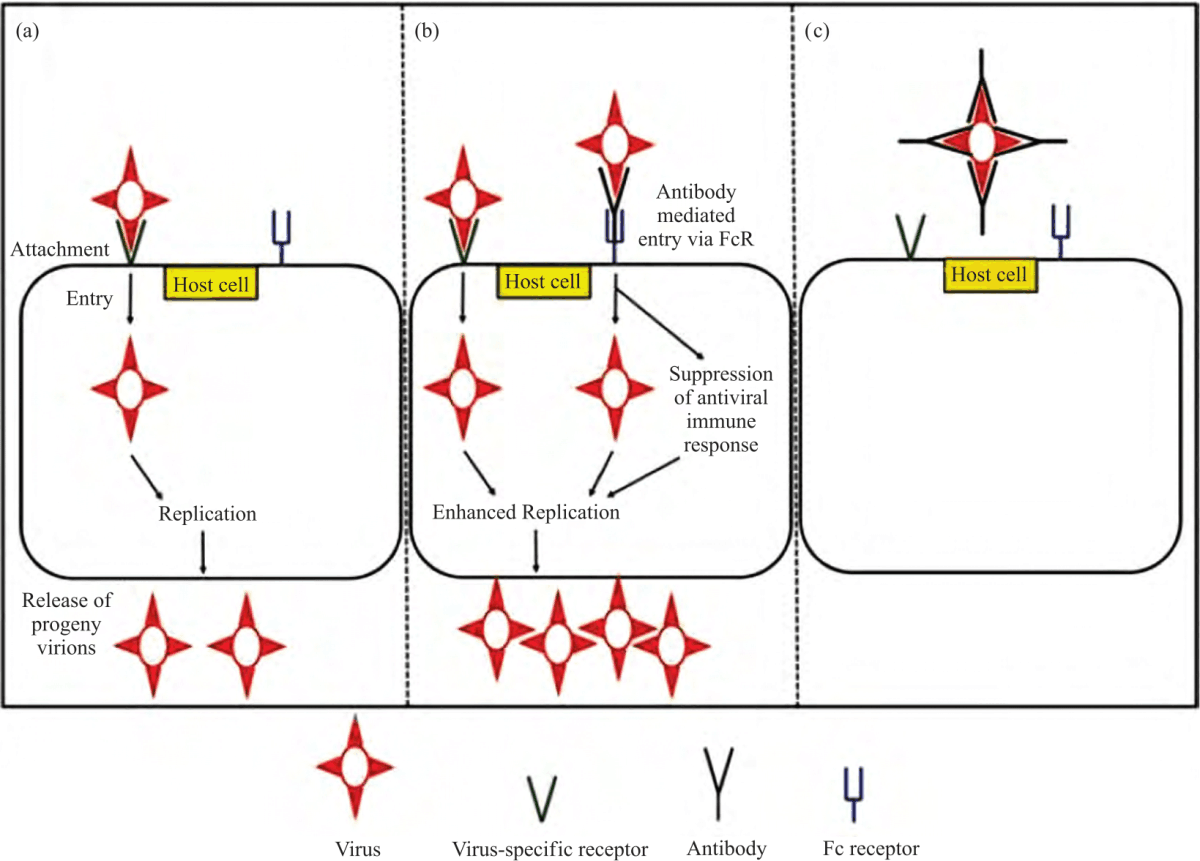
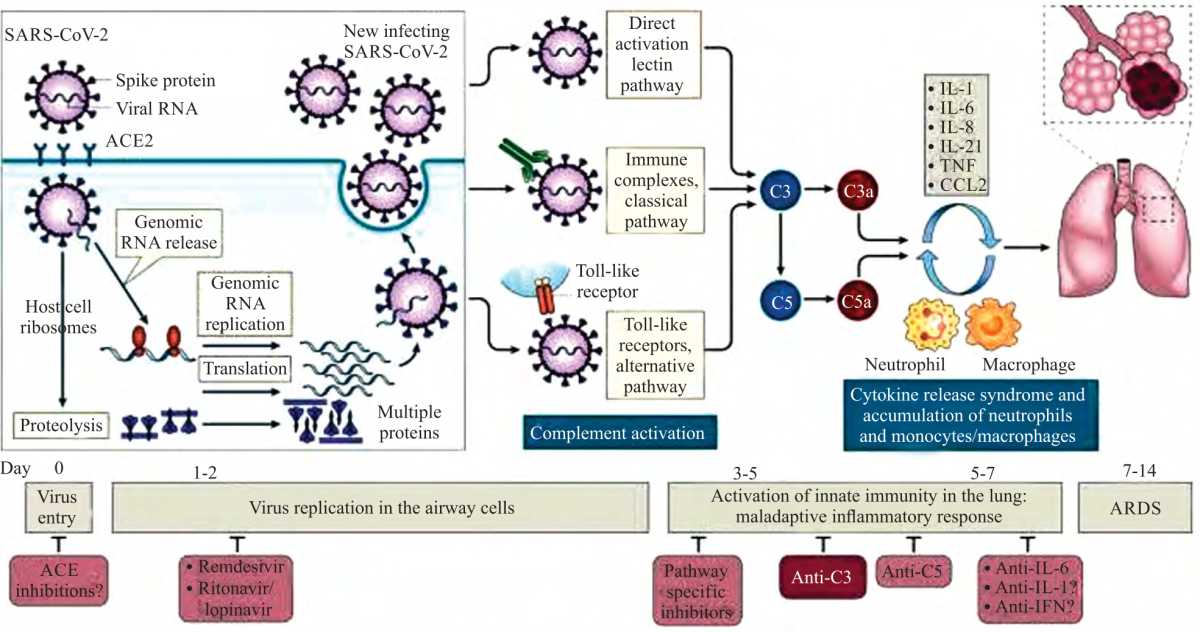
Review Article
Immune Response and Antibody-Dependent Mechanism Against Viral Infections: SARS-CoV-2 Vaccine as a Case Study
Department of Immunology, Institute of Biomedical Sciences, University of S�o Paulo, S�o Paulo, Brazil
LiveDNA: 234.32966
John Oluwafemi Teibo
Department of Biochemistry and Immunology, Ribeir�o Preto Medical School, University of S�o Paulo, Ribeir�o Preto, S�o Paulo, Brazil
Olayinka Anthony Adebayo
Department of Biochemistry, Molecular Drug Metabolism and Toxicology, University of Ibadan, Ibadan, Oyo State, Nigeria
Opeyemi Abigail Akinfe
Department of Biochemistry, Cancer and Molecular Biology Laboratory, University of Ibadan, Ibadan, Oyo State, Nigeria
Olaide Damilola Olagoke
Department of Biochemistry, Molecular Drug Metabolism and Toxicology, University of Ibadan, Ibadan, Oyo State, Nigeria