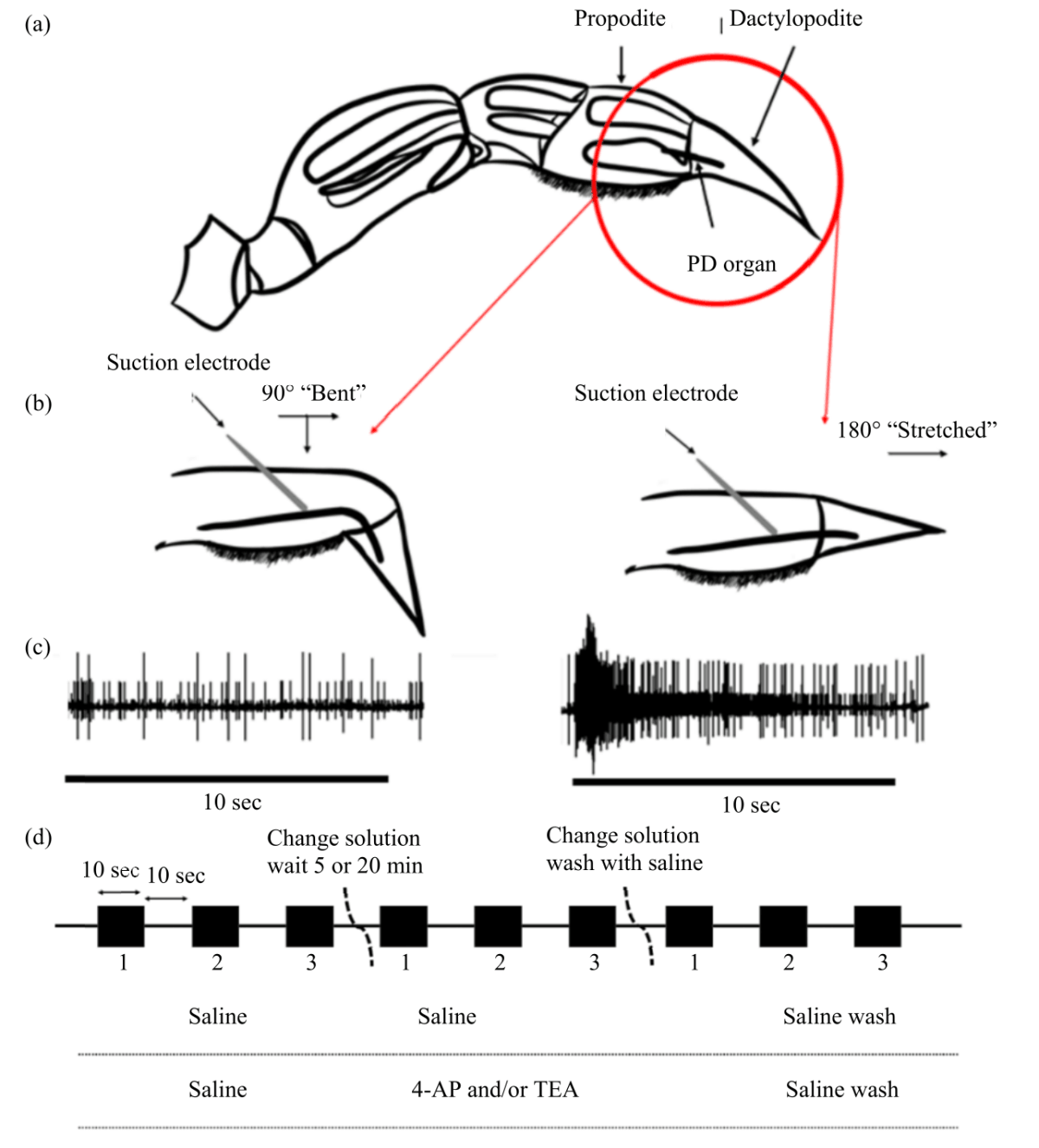
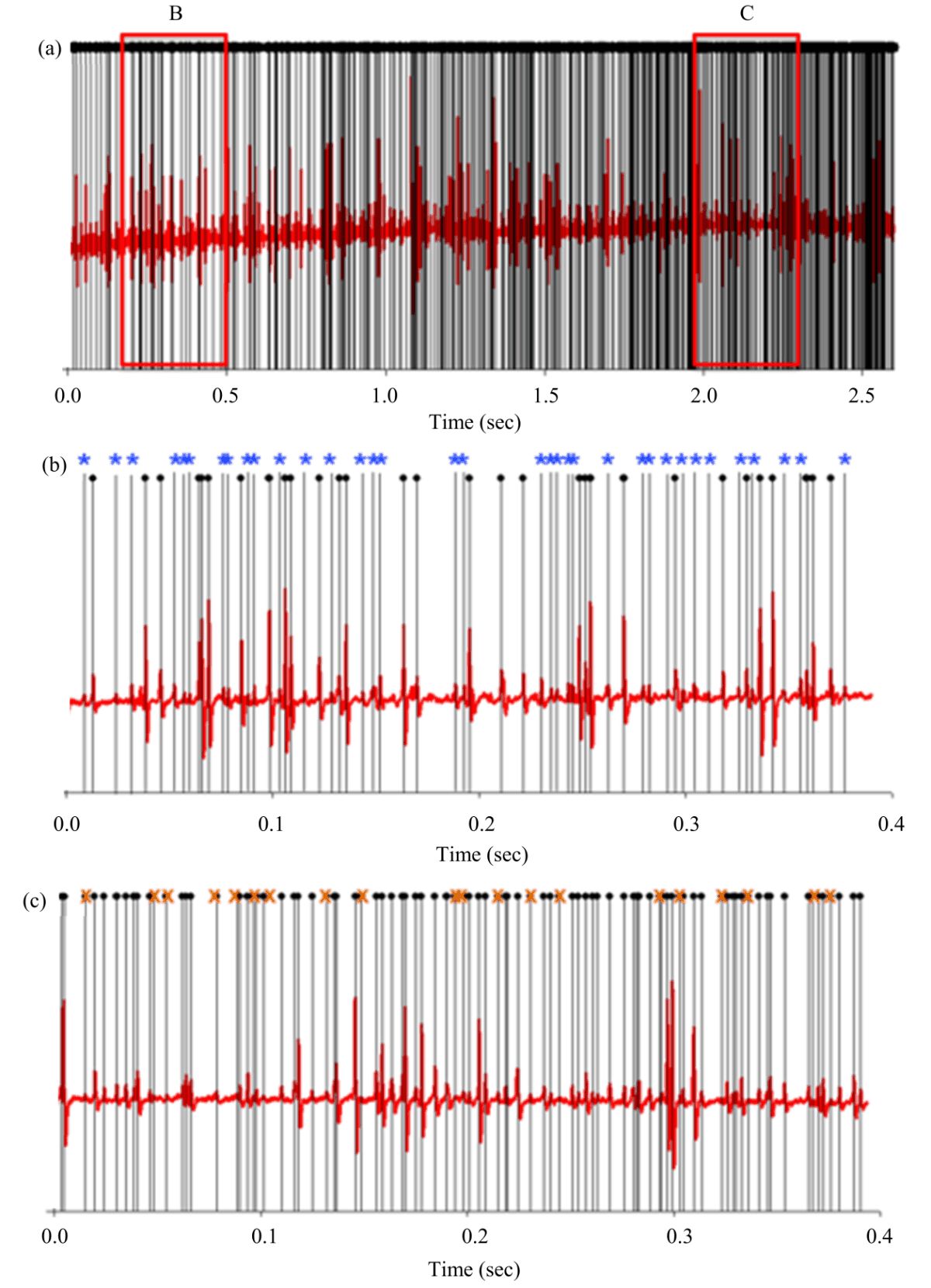
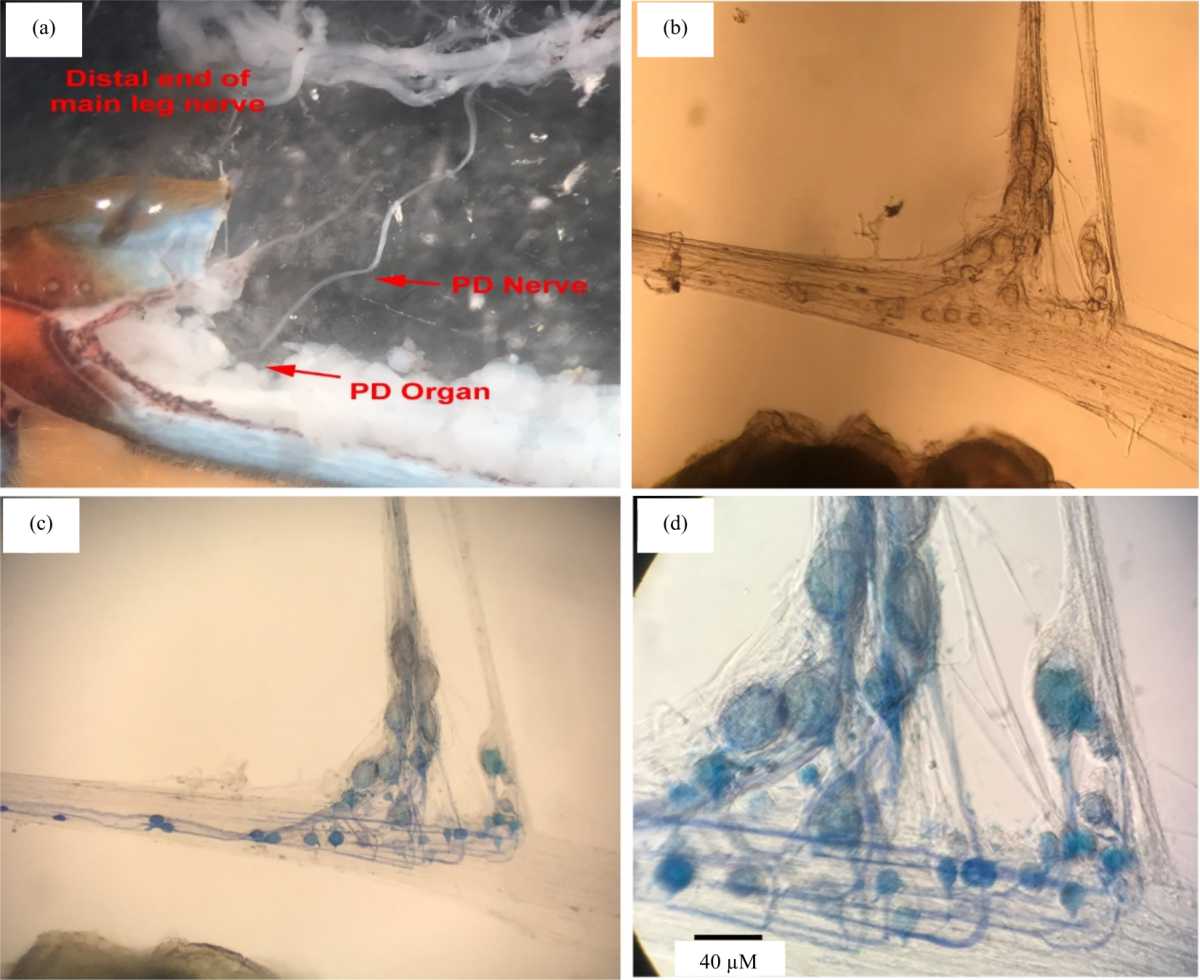
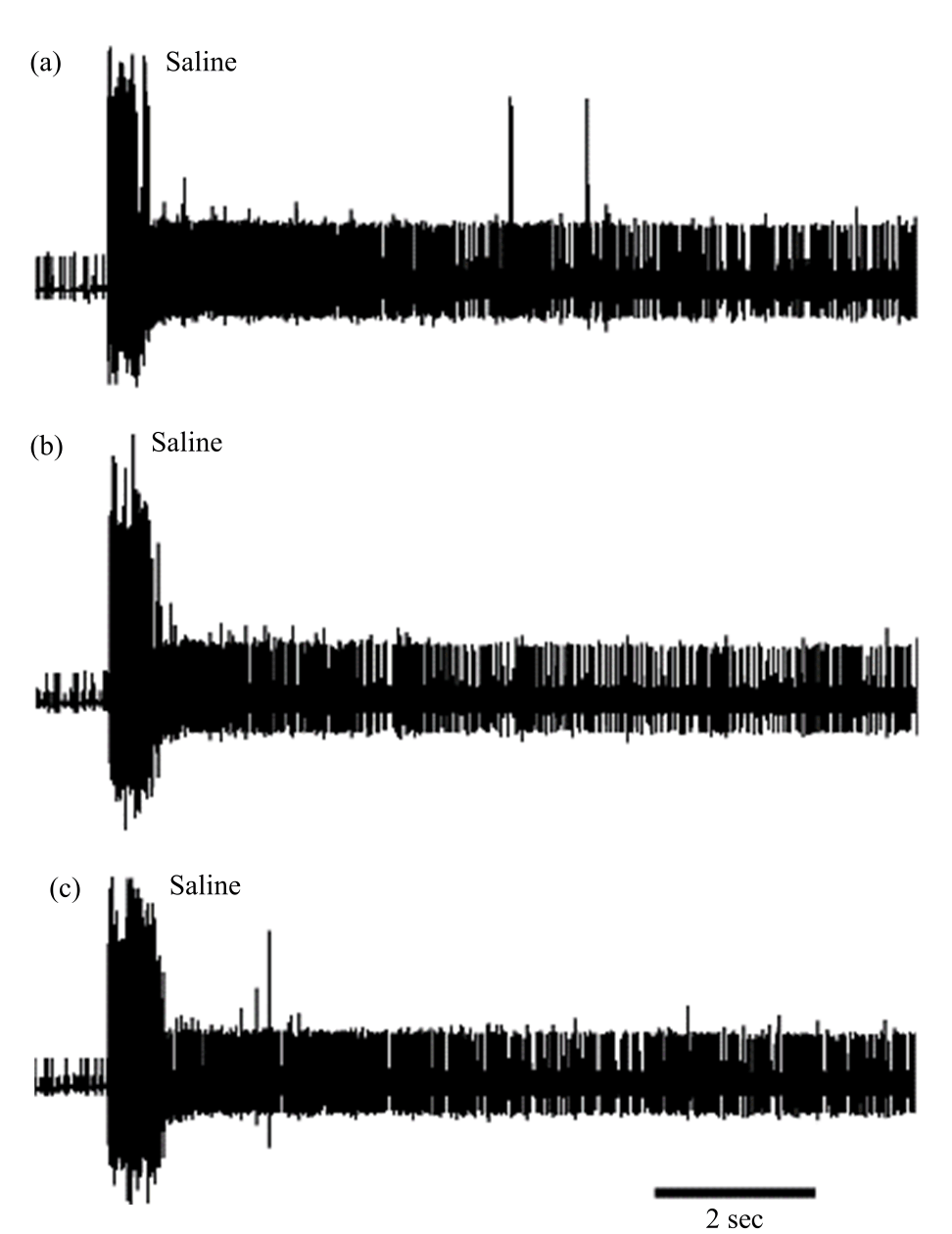
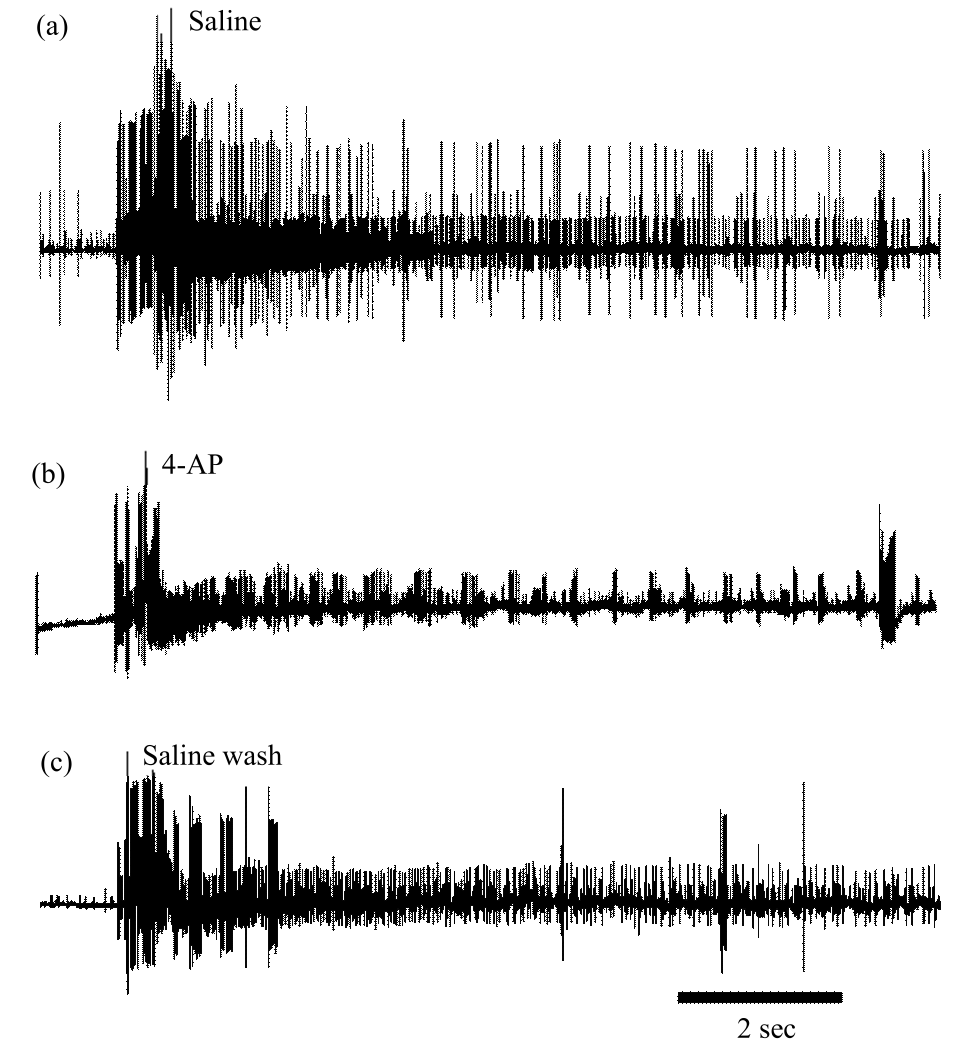
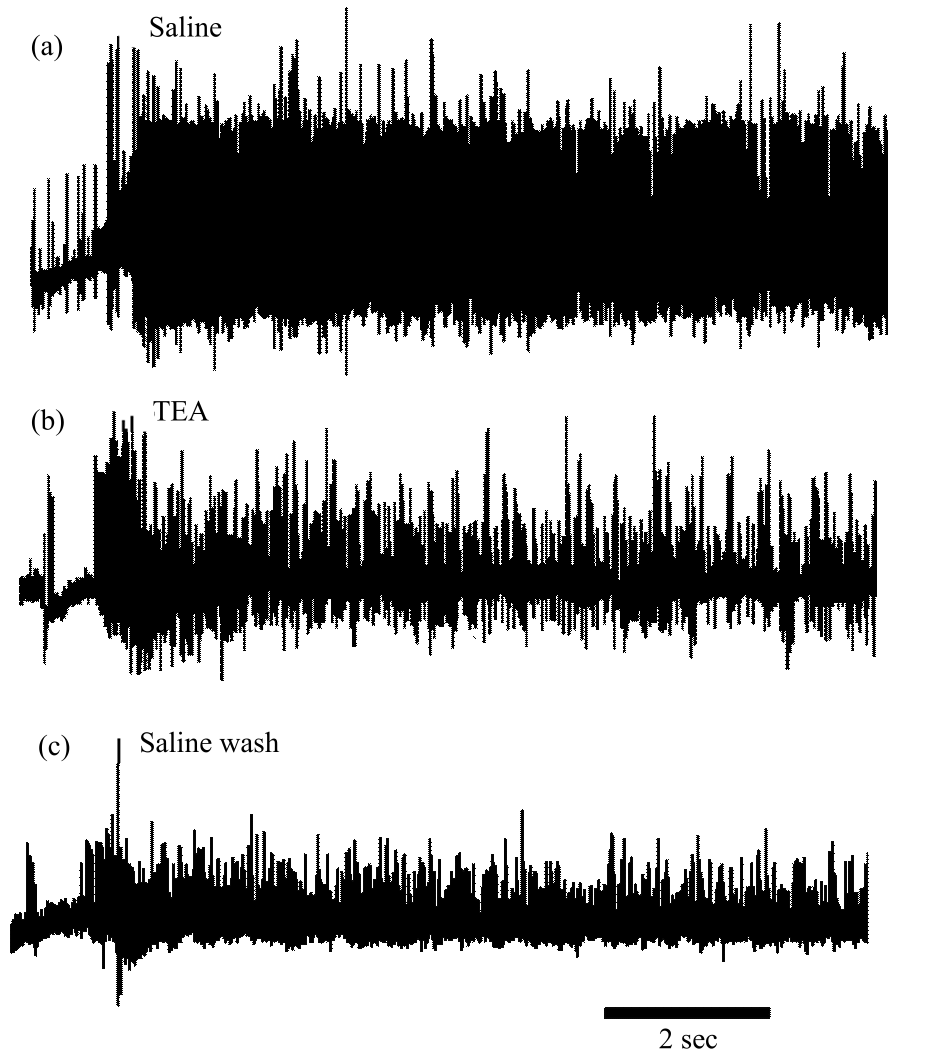
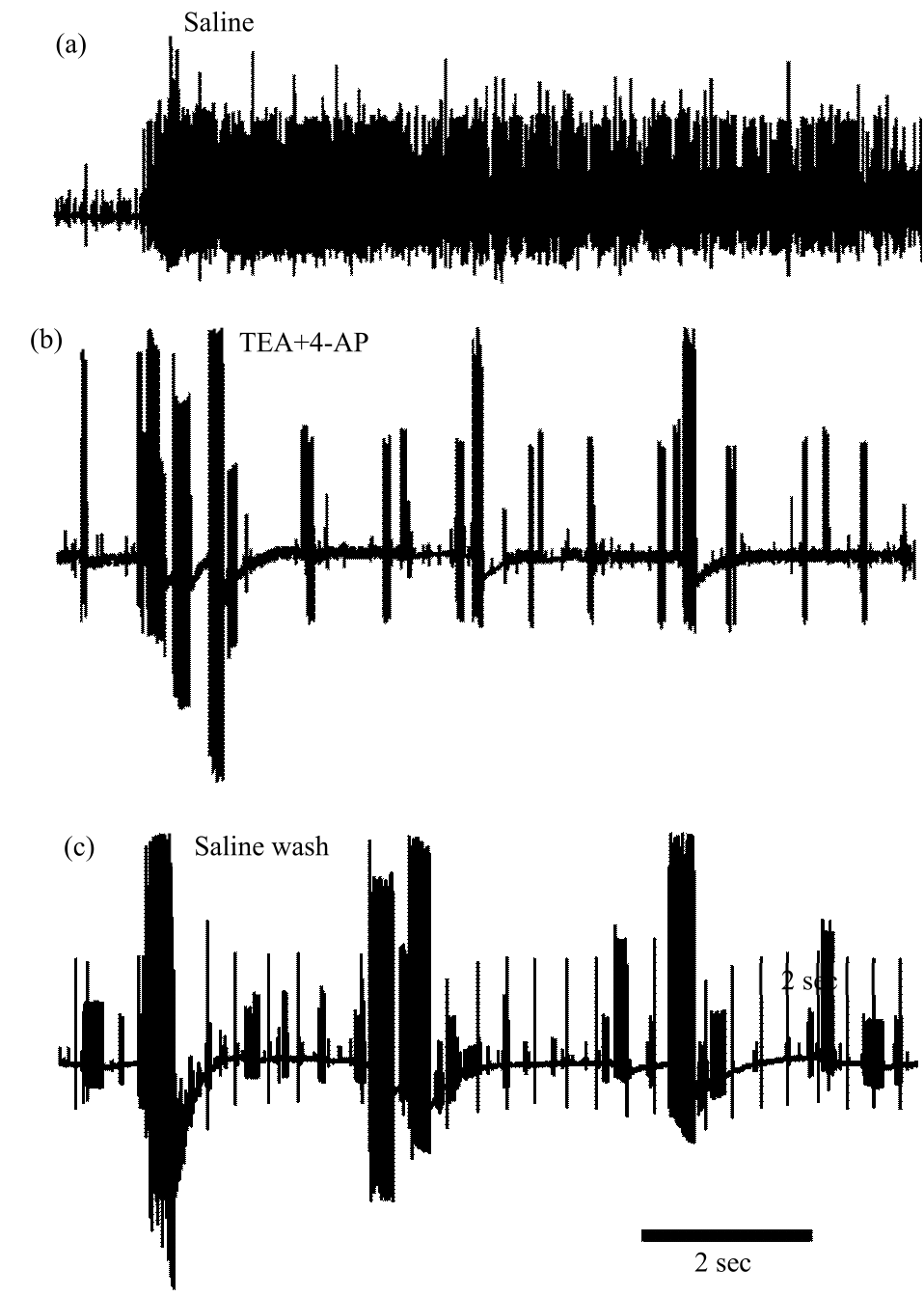
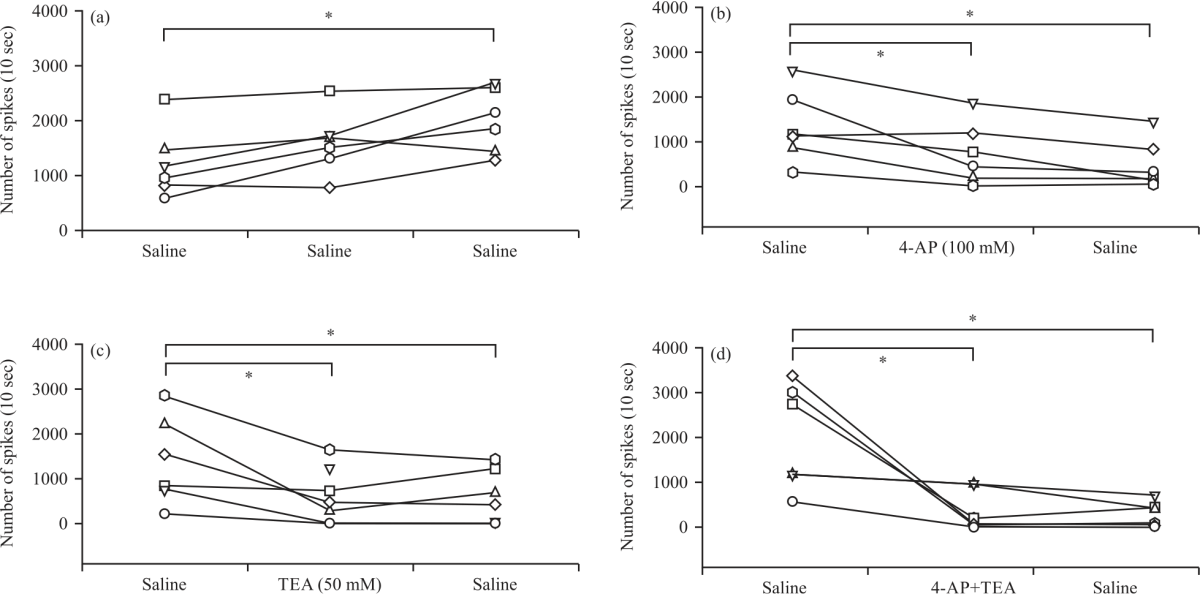
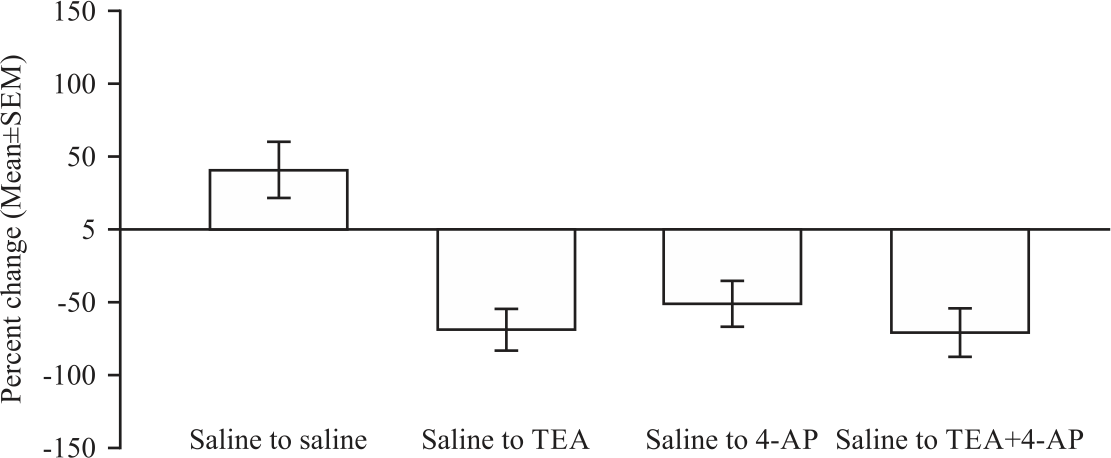
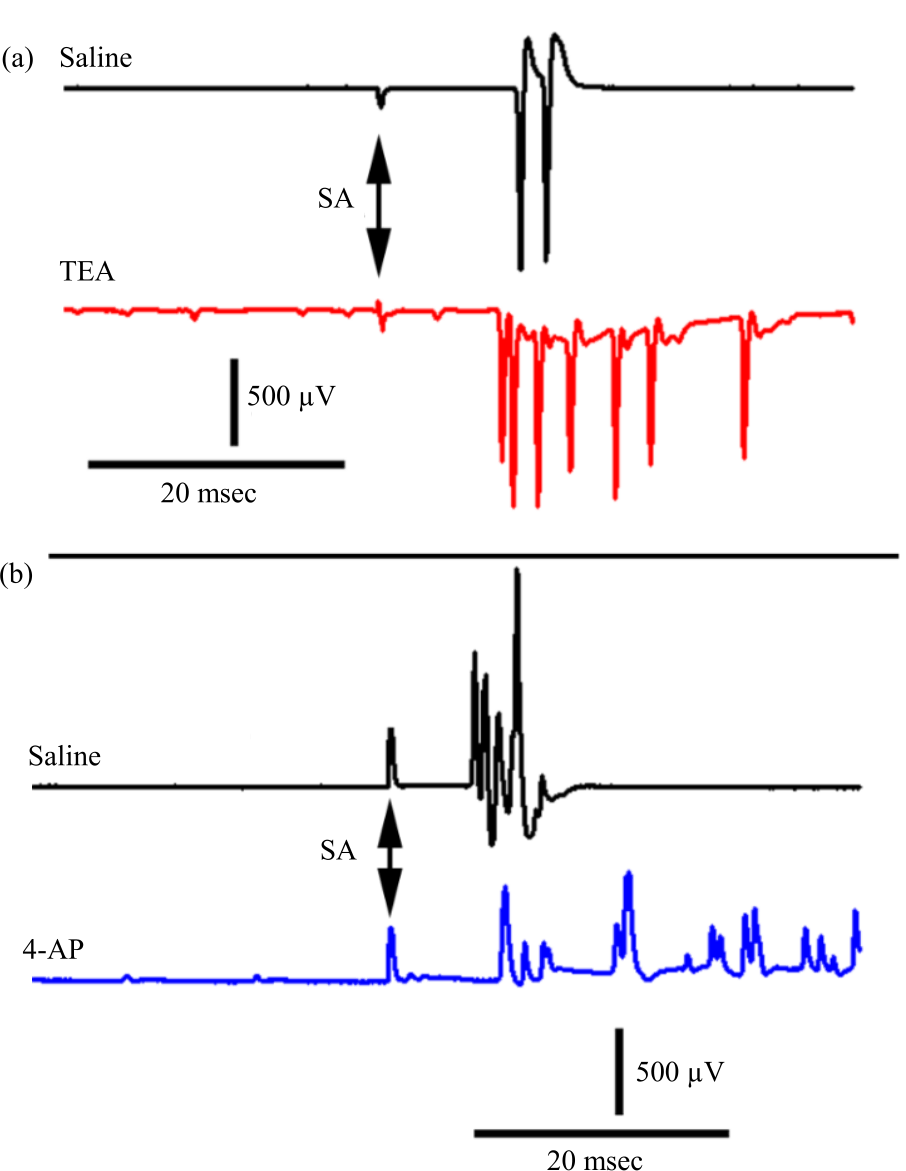
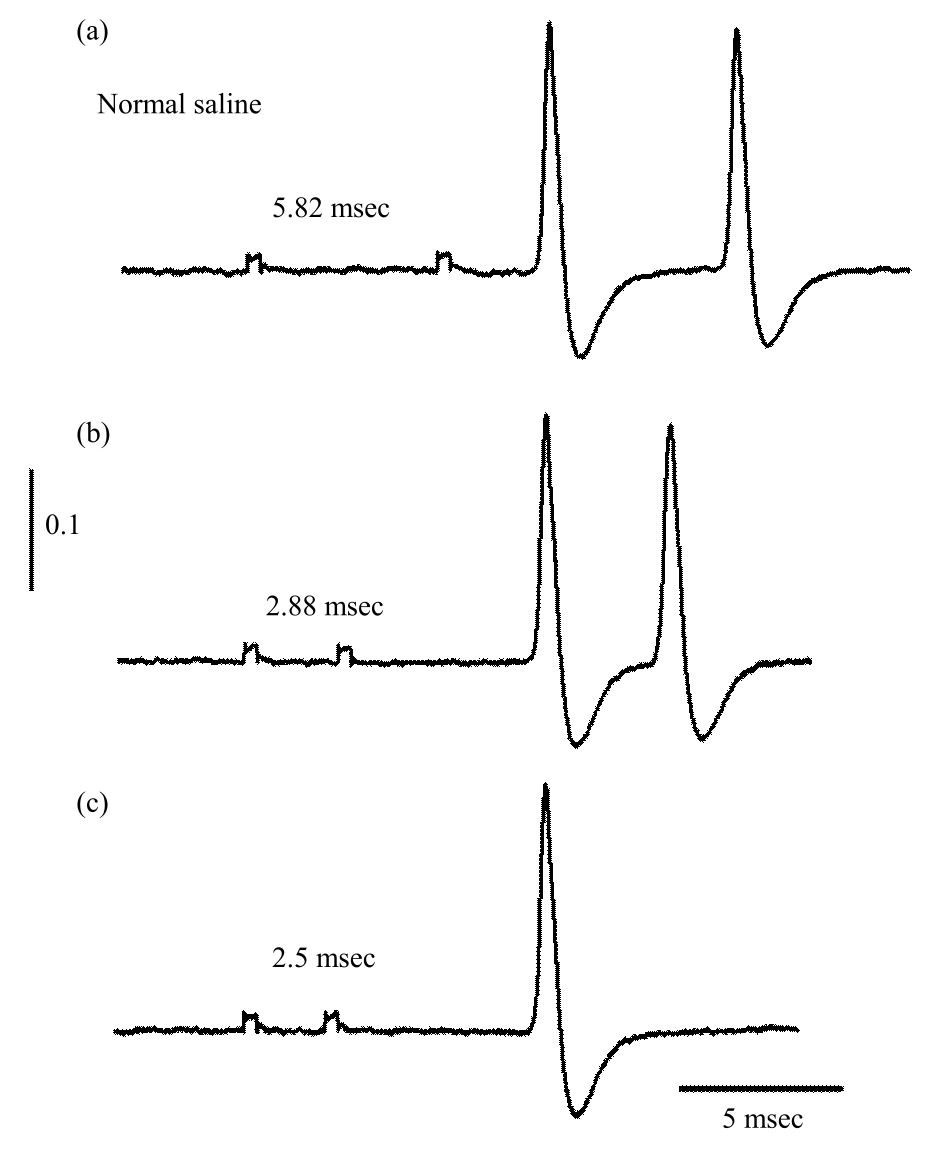
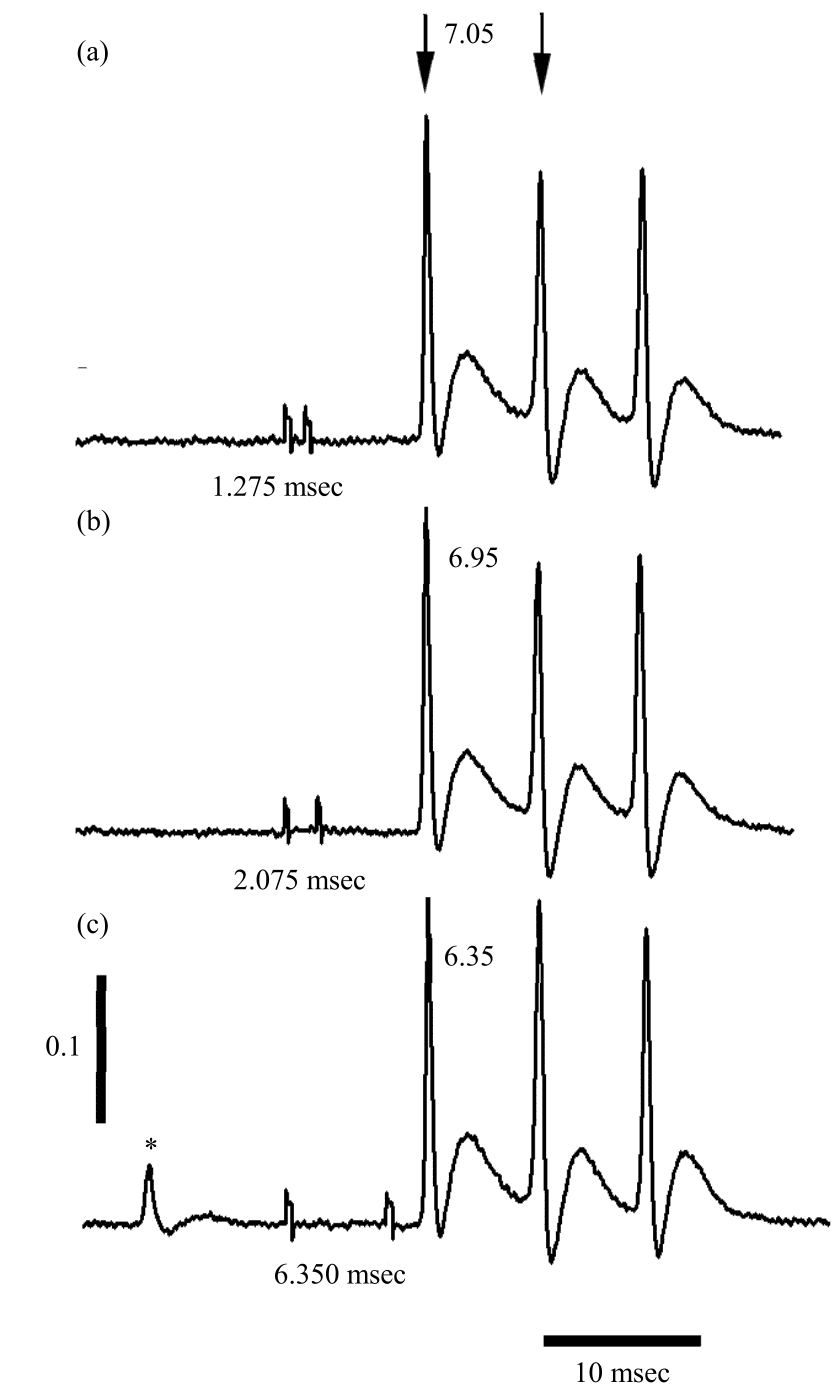
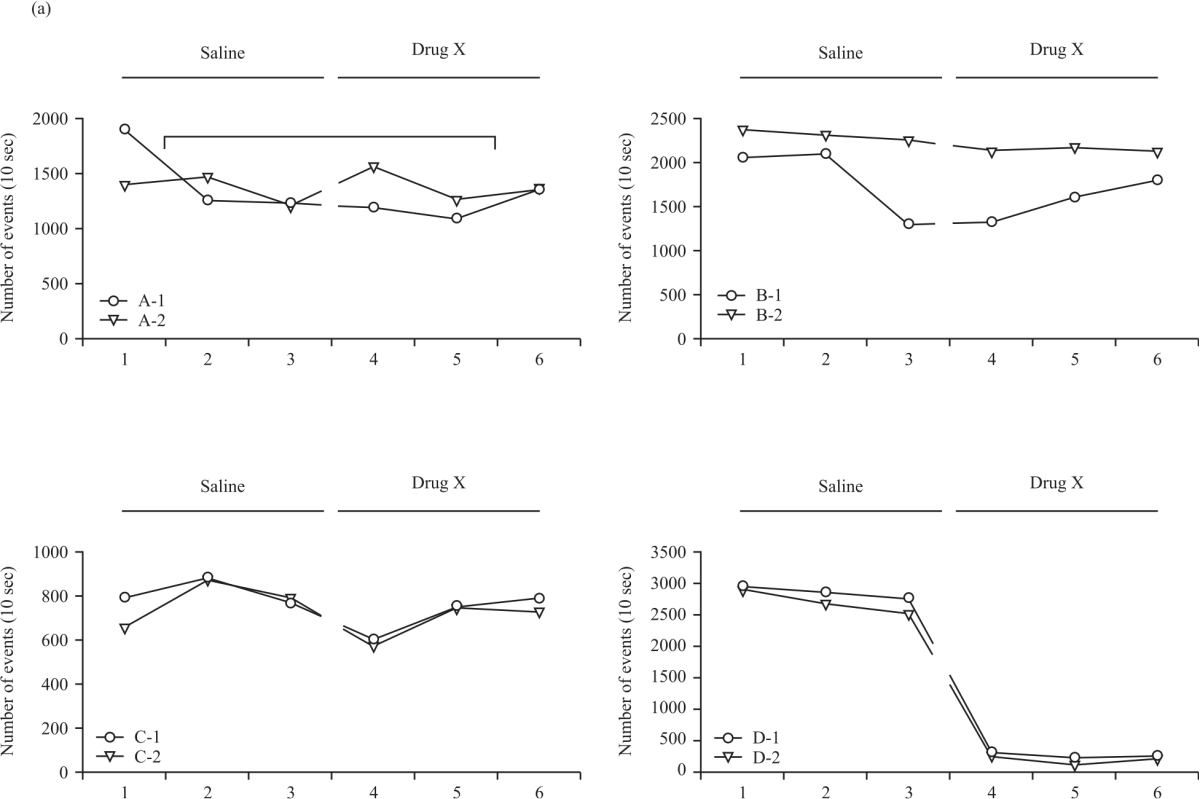
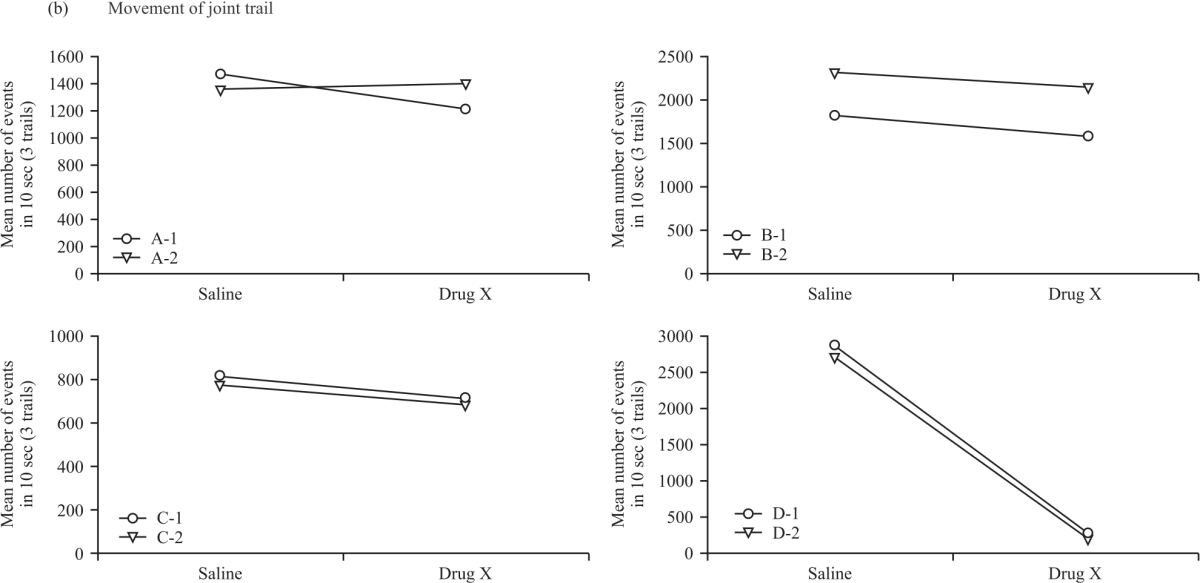
Research Article
Effect of TEA and 4-AP on Primary Sensory Neurons in a Crustacean Model
Department of Biology, Eastern Kentucky University, Richmond 40475, Kentucky, United States of America
Devan E. Atkins
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Kimberly L. Bosh
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Grace W. Breakfield
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Sydney E. Daniels
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Makayla J. Devore
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Hailey E. Fite
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Landys Z. Guo
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Danielle K.J. Henry
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Alana K. Kaffenberger
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Katherine S. Manning
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Tatum E. Mowery
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Cecilia L. Pankau
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Malina E. Serrano
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Yamaan Shakhashiro
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Ruth A. Ward
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Aubrey H. Wehry
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
Robin L. Cooper
Department of Biology, University of Kentucky, Lexington 40506, Kentucky, United States of America
LiveDNA: 1.523