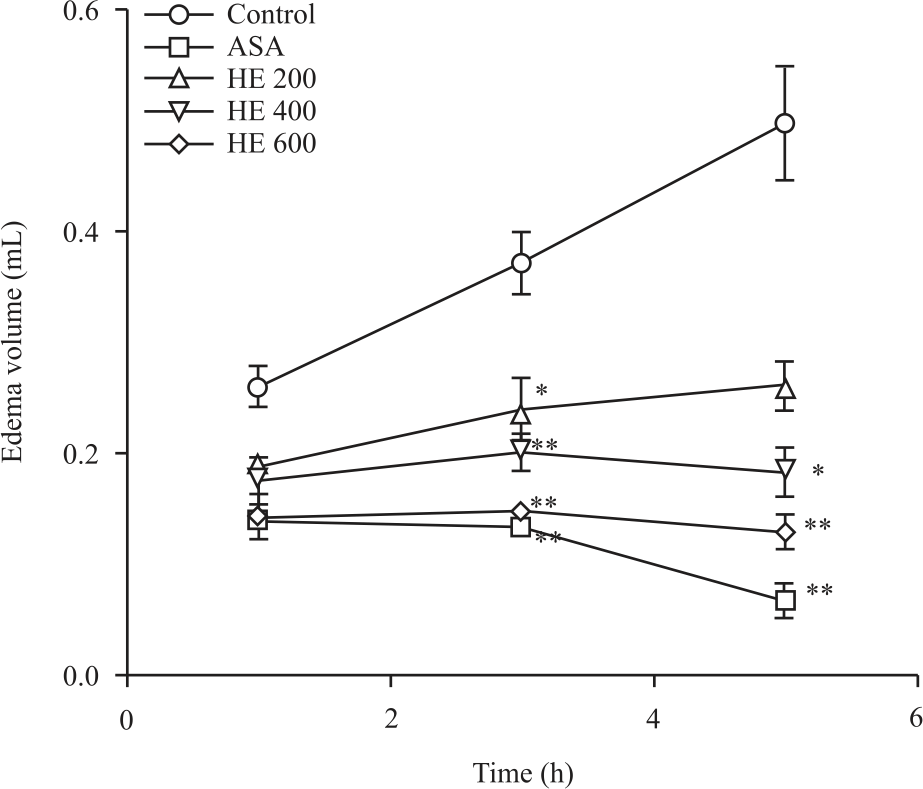
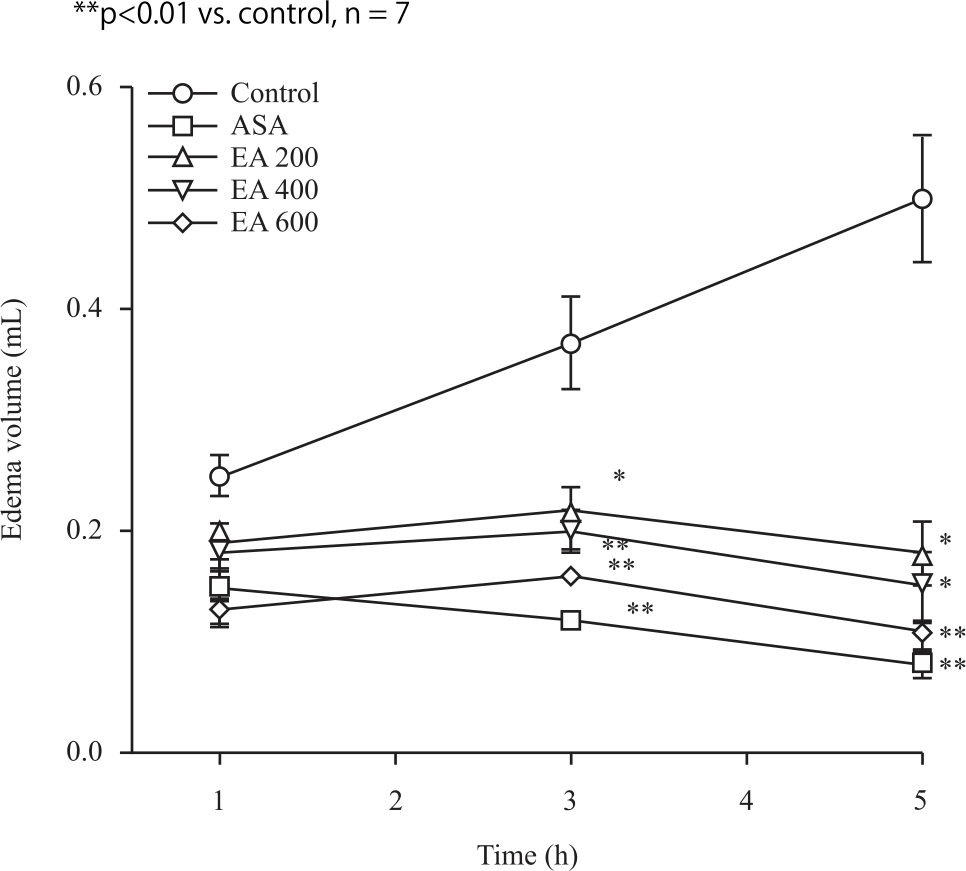
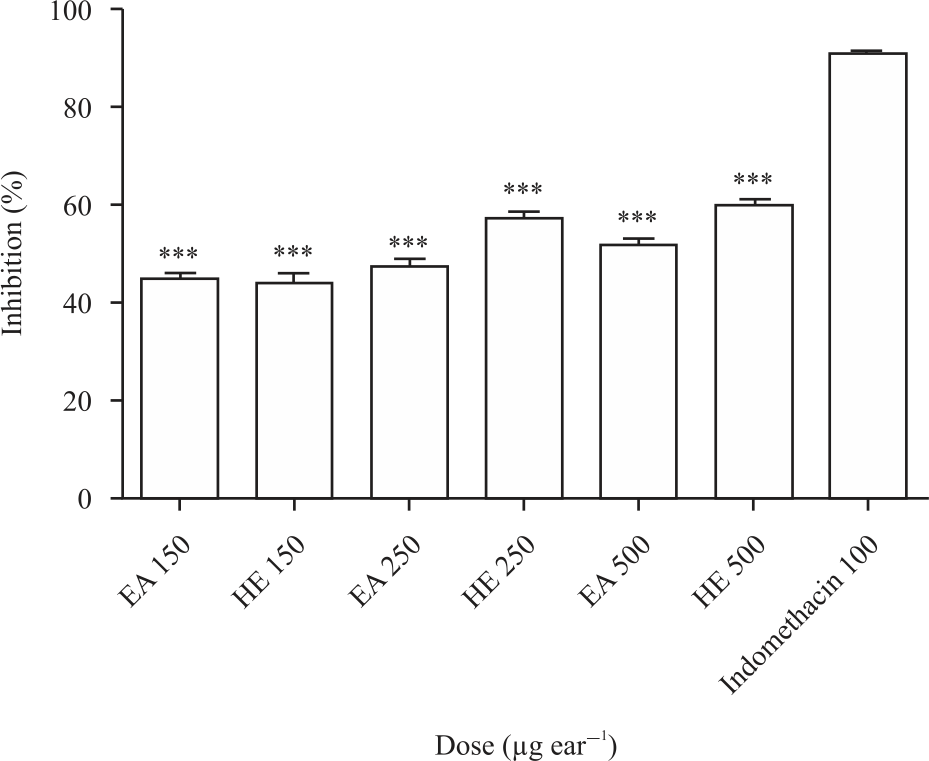
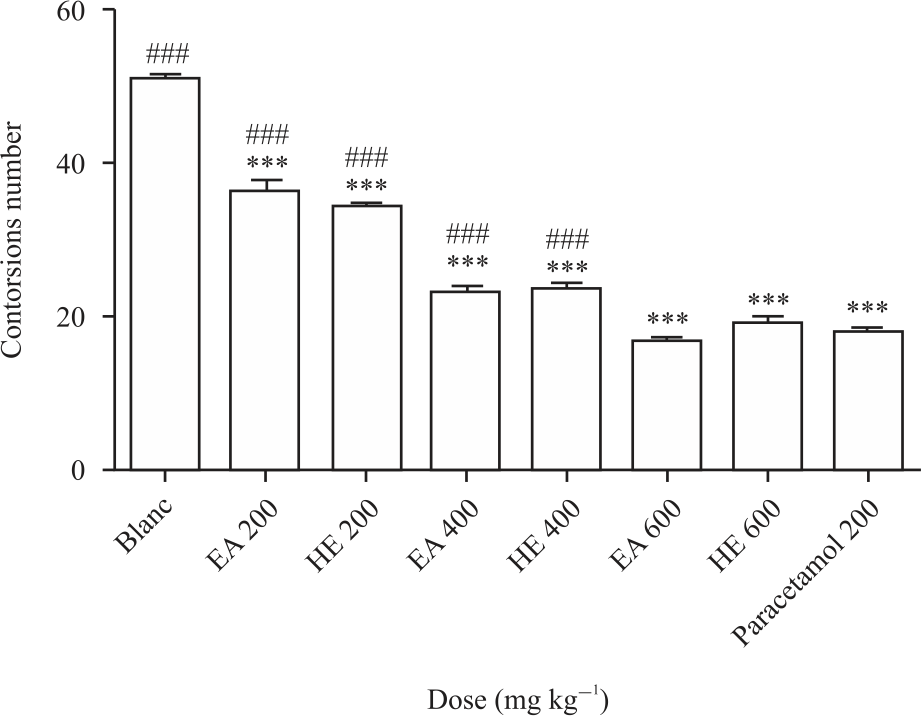
Research Article
Anti-Inflammatory Activity of Extracts from Parkia biglobosa (Jacq.) R.Br. Ex G.Don. (Fabaceae-Mimosoideae) Trunk Bark
Department of Traditional Medicine-Pharmacopeia/Pharmacy, Research Institute of Health Sciences (IRSS/CNRST), 03 BP 7047 Ouagadougou 03, Burkina Faso
LiveDNA: 226.34964
Constantin Atchade
Department of Traditional Medicine-Pharmacopeia/Pharmacy, Research Institute of Health Sciences (IRSS/CNRST), 03 BP 7047 Ouagadougou 03, Burkina Faso
Tata Kadiatou Traore
Department of Traditional Medicine-Pharmacopeia/Pharmacy, Research Institute of Health Sciences (IRSS/CNRST), 03 BP 7047 Ouagadougou 03, Burkina Faso
Benjamin Ouedraogo
Department of Traditional Medicine-Pharmacopeia/Pharmacy, Research Institute of Health Sciences (IRSS/CNRST), 03 BP 7047 Ouagadougou 03, Burkina Faso
Boly A.G. Laurent
Department of Traditional Medicine-Pharmacopeia/Pharmacy, Research Institute of Health Sciences (IRSS/CNRST), 03 BP 7047 Ouagadougou 03, Burkina Faso
W.L.M. Esther Kabre
Department of Traditional Medicine-Pharmacopeia/Pharmacy, Research Institute of Health Sciences (IRSS/CNRST), 03 BP 7047 Ouagadougou 03, Burkina Faso
Jules Yoda
Department of Traditional Medicine-Pharmacopeia/Pharmacy, Research Institute of Health Sciences (IRSS/CNRST), 03 BP 7047 Ouagadougou 03, Burkina Faso
Felix B. Kini
Department of Traditional Medicine-Pharmacopeia/Pharmacy, Research Institute of Health Sciences (IRSS/CNRST), 03 BP 7047 Ouagadougou 03, Burkina Faso
Marius Lompo
Department of Traditional Medicine-Pharmacopeia/Pharmacy, Research Institute of Health Sciences (IRSS/CNRST), 03 BP 7047 Ouagadougou 03, Burkina Faso
Moussa Ouedraogo
Laboratory of Drug Development, Doctoral School of Health, University Joseph KI ZERBO, 03 BP 7021, Ouagadougou 03, Burkina Faso
Sylvin Ouedraogo
Department of Traditional Medicine-Pharmacopeia/Pharmacy, Research Institute of Health Sciences (IRSS/CNRST), 03 BP 7047 Ouagadougou 03, Burkina Faso