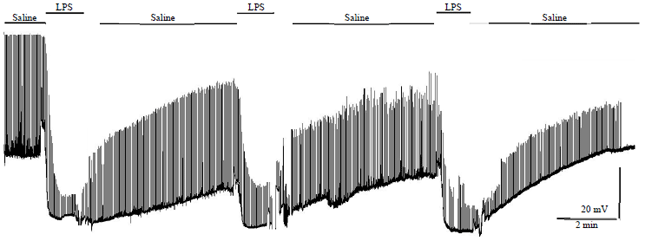
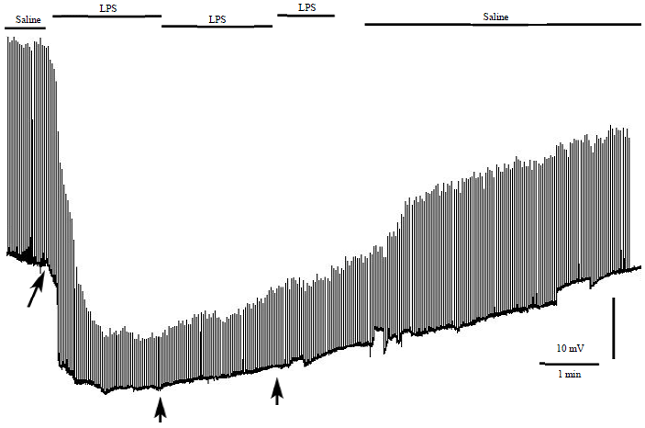
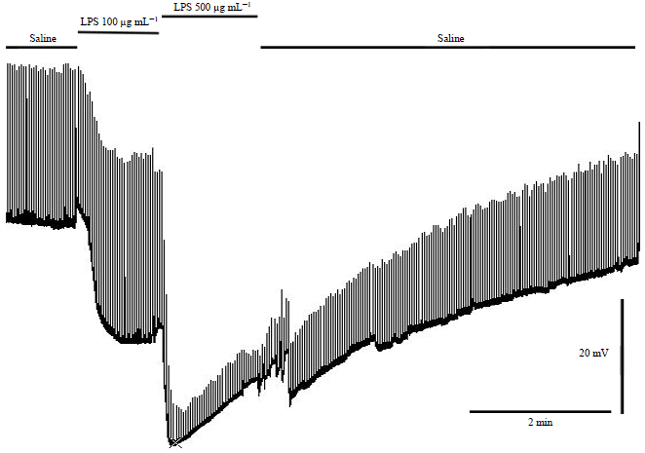
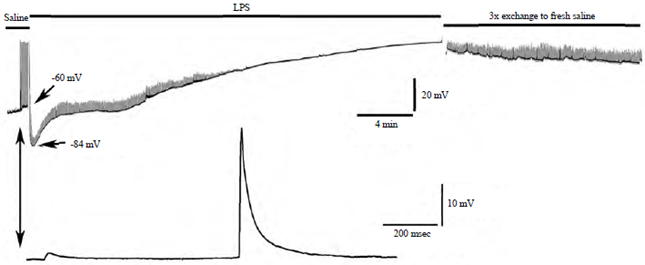
Research Article
Repetitive Exposure to Bacterial Endotoxin LPS Alters Synaptic Transmission
Department of Biology, Center for Muscle Biology, University of Kentucky, Lexington, KY, 40506-0225, USA
Abigail Greenhalgh
Department of Biology, Center for Muscle Biology, University of Kentucky, Lexington, KY, 40506-0225, USA
Robin L. Cooper
Department of Biology, Center for Muscle Biology, University of Kentucky, Lexington, KY, 40506-0225, USA
LiveDNA: 1.523