Research Article
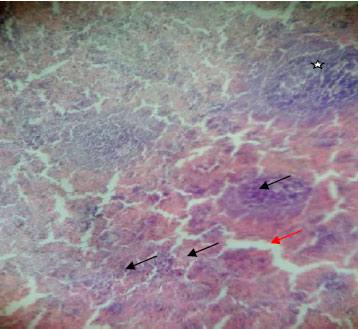
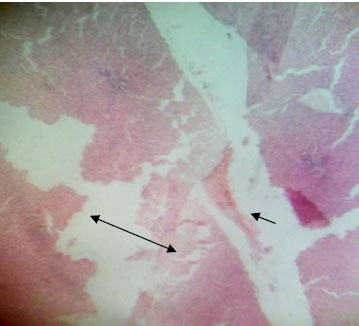
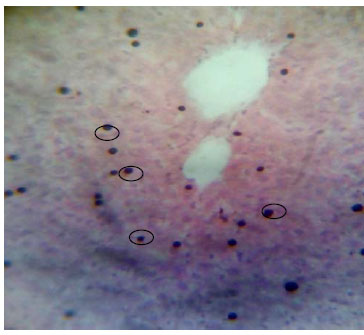
Determination of Some Phyto-constituents Present in Terminalia catappa Endocarp Flour and Biochemical Evaluation of its Ethanolic Extract on Hepatic Indices in Male Wistar Rats
Department of Biochemistry, Michael Okpara University of Agriculture, Umudike, P.M.B. 7267 Umuahia Abia State, Nigeria
LiveDNA: 234.20815
Achi Ngozi K
Department of Biochemistry, Michael Okpara University of Agriculture, Umudike, P.M.B. 7267 Umuahia Abia State, Nigeria
Egbuonu Anthony C. Cemaluk
Department of Biochemistry, Michael Okpara University of Agriculture, Umudike, P.M.B. 7267 Umuahia Abia State, Nigeria
Egu Elizabeth Uchechi
Department of Human Nutrition and Dietetics, Michael Okpara University of Agriculture, Umudike, P.M.B. 7267 Umuahia Abia State, Nigeria