Research Article
Protective Effect of Melatonin on Gentamicin Induced Hepatotoxicity in Rats
Department of Biology, Faculty of Science, Princess Nourah Bint Abdulrahman University, Riyadh, Kingdom of Saudi Arabia
LiveDNA: 966.16177
Aminoglycoside antibiotics are commonly used for the treatment of severe Gram-negative bacterial infections by inhabiting bacterial protein synthesis, therefore the most widely used drug in this category is gentamicin (GM)1. Although aminoglycosides have beneficial effects, they are also known for their nephrotoxicity effects2,3.
The liver is an important organ in the body; it controls an extensive array of biochemical and metabolic functions, removes harmful substances from the body, that would be injurious if they accumulate and eliminates drug metabolites4,5. It also maintains body homeostasis6, metabolism and biotransformation of drugs and xenobiotics that targets the liver7.
Hepatotoxicity induced by gentamicin in experimental animals were reported6-14. Gentamicin enhances the oxidative stress and generation of free radicals and causes inhibition of the antioxidant defense system in the liver. It suppresses the non-enzymatic and enzymatic antioxidants, which leads to an over production of Reactive Oxygen Species (ROS). This will cause damage to membrane lipids, proteins and nucleic acids, which leads to liver toxicity, dysfunction and injury7.
Many natural products have been used to protect the body from toxicities caused by drugs. Generally, herbs are considered effective and safe against several human diseases15. Natural antioxidants enhance the internal antioxidants defenses from ROS and improve the balance by neutralizing the reactive species16.
Melatonin N-acetyl-5-methoxytryptamine is a secretory product of the pineal gland that participates in many physiological functions, including seasonal reproduction, immunity and circadian rhythms17. Several documents reported melatonin’s protective actions in different models of oxidative stress in response to its direct free radical scavenger activity and its indirect antioxidant properties18-27. The biological membranes are easily crossed by Melatonin, hence reaching all components of the cell28, melatonin protects against gentamicin-induced nephrotoxicity29,30. The use of melatonin combined with GM may help in reducing some of its side effects. It may open new aspects in natural therapy.
The present study was designed to investigate the protective effects of melatonin against hepatotoxicity and oxidative stress in rats treated with GM.
The study was carried out at Princess Nourah Bint Abdulrahman University, Riyadh, Kingdom of Saudi Arabia, 2016.
Experimental design: Male albino rats weighed about 150-200 g were used. All animals were husbandry and treated accordance with the public Health Guide for the Care and Use of Laboratory Animals31, they were randomly divided into four equal groups, five animals in each group: (1) Control group: Rats didn’t receive any treatment. (2) Gentamycin (GM) group: Rats were intraperitoneally injected daily with GM (40 mg kg–1)11 for seven days. (3) Melatonin group: Rats were intraperitoneally injected daily with melatonin (10 mg kg–1) for 10 day’s29. (4) Melatonin and GM group: Rats were intraperitoneally injected daily with melatonin (10 mg kg–1) for three days then continued seven days with the GM (40 mg kg–1) together with the melatonin. All animals were sacrificed 24 h after the last injection. Blood was collected to obtain serum which was stored at -20°C until the determination of total lipid, triglyceride, phospholipids, total cholesterol, High Density Lipoprotein (HDL), Low Density Lipoprotein (LDL), alanine aminotransferase (ALT), aspartate aminotransferase (AST) and total bilirubin using kits purchased from Biomerieux, France.
Small samples of liver were collected and weighed then homogenized in ice-cold tris-HCl buffer pH 7.5, the homogenate was centrifuged at 3000 rpm for 15 min in a refrigerated high-speed centrifuge at 4°C and resulting supernatant was stored at -20°C until the determination of lipid peroxidation products (Malondialdehyde (MDA)), it was estimated as described by Ohkawa et al.32, glutathione (GSH) content was measured as described by Prins and Loos33, glutathione peroxidase (GSH-Px) was assayed by using ready kits from Randox Laboratories Ltd. UK, glutathione-S-transferase (GST) was measured according to Habig et al.34, catalase (CAT) activity was assayed as described by Bock et al.35, hydroxyproline was measured as described by Bergman et al.36 and protein carbonyl was estimated according to Levine et al.37.
Statistical analysis: Statistical analysis was performed with SPSS 10.0 package (SPSS 10 for Windows, SPSS Inc., Chicago, IL, USA.). The values reported are Means±SEM (number of rats). Differences between group means were estimated using one-way analysis of variance (ANOVA)38 and were considered significant when p<0.05.
The effect of gentamicin (GM) and melatonin treatment on serum total bilirubin, aspartate aminotransferase (AST) and alanine aminotransferase (ALT) activities.
There is an increase in serum total bilirubin, AST and ALT activities in the GM group compared to the control group (Table 1).
| Table 1: | Protective effect of melatonin (10 mg kg–1) against gentamicin (40 mg kg–1) on biochemical blood parameters levels in serum |
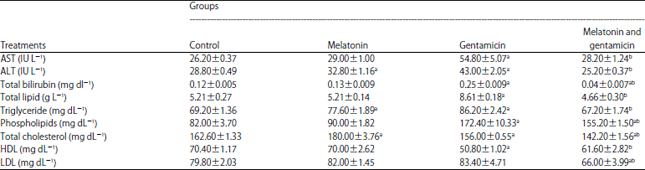 | |
AST: Aspartate aminotransferase, ALT: Alanine aminotransferase , HDL: High density lipoprotein, LDL: Low density lipoprotein. Values are expressed as Means±SE (n = 5), a Significant compared to the control group (p<0.05), b Significant compared to the gentamicin group (p<0.05) | |
| Table 2: | Protective effect of melatonin (10 mg kg–1) against gentamicin (40 mg kg–1) on oxidative stress biomarkers in liver tissues of male rats |
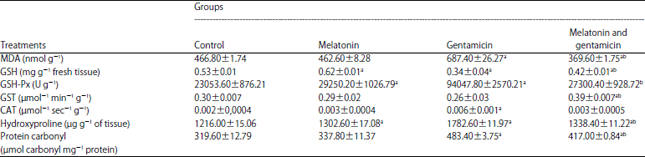 | |
MDA: Malondialdehyde, GSH: Glutathione, GSH-Px: Glutathione peroxidase, GST: Glutathione-S-transferase, CAT: Catalase. Values are expressed as Means±SE (n = 5), a Significant compared to the control group (p<0.05), b Significant compared to the gentamicin group (p<0.05) | |
Pretreatment of animals with melatonin significantly (p<0.05) reduced high levels of serum total bilirubin, also serum levels of AST and ALT were in normal range as compared with the control group.
The effect of GM and melatonin treatment on serum total lipid, triglyceride, phospholipids, total cholesterol, High Density Lipoprotein (HDL) and Low Density Lipoprotein (LDL).
The injection with GM caused significant (p<0.05) increase in total lipid, triglyceride and phospholipids compared with the control group. while there was a significant (p<0.05) decrease in total cholesterol and HDL compared with the control. The combination of melatonin and GM, total lipid, triglyceride and HDL were in normal range as compared to the control group. Also, there was a significant (p<0.05) decrease in phospholipids, total cholesterol and LDL as compared to the GM group (Table 1).
The effect of GM and melatonin treatment on liver malondialdehyde (MDA), glutathione (GSH), glutathione peroxidase (GSH-Px), glutathione-S-transferase (GST), catalase (CAT), hydroxyproline and protein carbonyl.
There is a significant (p<0.05) increase in MDA concentration, GSH-Px, CAT, hydroxyproline and protein carbonyl in rats treated with GM when compared to the control group, it also showed a decrease in liver GSH content as shown in Table 2. While the combination of melatonin and GM prevented the normal activity of liver GSH-Px and CAT. There was a decrease in MDA, hydroxyproline, protein carbonyl and an increase in GSH content, GST activity as compared to the GM group.
Gentamicin’s (GM) clinical benefits are restricted due to its nephrotoxicity, evidence in vitro and vivo indicates that reactive oxygen metabolites are important mediators of gentamicin nephrotoxicity. Therefore, amelioration of nephrotoxicity would enhance the clinical use of GM. Liver and kidneys are vital organs actively involved in metabolic homeostasis, which generates several Reactive Oxygen Species (ROS) reactions. Therefore, these organs are more susceptible to oxidant injury due to their metabolic rate39-42.
In this study, GM treatment caused liver injury in rats, as indicated by significant (p<0.05) increases in alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities in serum, our results are in line with4,7,8,43. In general, serum levels of transaminases are sensitive markers of liver function and their levels are increased in serum, thus released in blood by altering the permeability of hepatocyte membranes44. In this study, pretreatment of animals with melatonin serum levels of AST and ALT were in normal range as compared to normal group.
The GM administration causes significant (p<0.05) increase in serum level of liver function markers such as total lipids, phospholipids, triglycerides and total bilirubin as compared to the control group, indicating hepatic dysfunction, due to the involvement of oxidative stress and production of free radicals to hepatotoxicity caused by GM treatment. The various metabolic pathways of the liver might be influenced by GM, thereby enhancing the level of total cholesterol, triglycerides and total bilirubin. The same results have been reported in different studies8,44, that pretreatment of animals with melatonin decreased the concentration of liver function markers.
Rats treated with GM mediates the generation of ROS that play an important role in progression of hepatic and renal injuries, as well as an array of biomolecules such as membrane lipids, protein and nucleic acids, especially in mitochondria and lysosomes of renal tissues45. Peroxidation of polyunsaturated fatty acids on bio membranes is due to an increase in ROS propagation. The GM caused lipid peroxidation, cellular function impairment and led to necrosis45. In this study, a significant (p<0.05) increase was observed in the lipid peroxidation product (TBARS) with GM, our results are in line with many studies7,9,44. The index of endogenous lipid peroxidation are TBARS, an indirect evidence of increased free radical production46. The TBARS levels in this study were elevated in the liver of GM treated group and were significantly (p<0.05) reduced when administered with melatonin, the ability of melatonin to stimulate the synthesis of antioxidants enzyme will most likely lead to reduction of malondialdehyde (MDA) levels and increase the glutathione peroxidase (GSH-Px)47. Yanagida et al.48 confirmed that GM may cause oxidative damage in rat hepatic microsomes. Production of hydrogen peroxide in liver mitochondria will increase when treated with GM, which may induce ROS and cell damage49.
Hepatic glutathione (GSH) was lower in gentamicin-treated animals, the results of present study are in line with previous studies7,50. The GSH plays a significant role in removing many reactive species, it plays a major role in several cellular processes including xenobiotics detoxification and protection from ROS51-54. The GSH acts as a free radical scavenger, it protects the cellular system from the toxic effects of lipid peroxidation. The significant (p<0.05) recovery of GSH and glutathione-S-transferase (GST) levels in liver after treatment is an indication of the protective effect of melatonin in this study. Catalase, GSH and GST are biochemical signs of oxidative stress and antioxidant status.
Concentration of liver collagen is estimated by measuring hydroxyproline content in liver. Levels of hydroxyproline are markers of fibrotic degeneration, protein carbonyl groups are markers of protein oxidation by ROS, oxidative damage is a substrate for hepatic fibrogenesis, it is found in the liver of patients with alcohol addiction, hepatitis C virus infection, iron overload, or chronic cholestasis, it is also found in types of experimental liver fibrogenesis55-57. The GM administration causes significant (p<0.05) increase in hydroxyproline content and Protein carbonyl groups in liver. However, there was a significant (p<0.05) decrease in hydroxyproline content and Protein carbonyl groups in the liver when administered with melatonin, antioxidants have arisen as powerful antifibrotic factors, melatonin has a higher antioxidant efficiency when compared with vitamin E and GSH, which are known as powerful antioxidants58,59. Hence, melatonin might be proposed as therapeutic modalities in experimental models of hepatic fibrosis. In other models of liver injury, melatonin was shown to protect the liver by inhibiting oxidative damage60-62. In another study, treatment of the fibrotic rat in early stages with melatonin decreased MDA and increased hepatic antioxidant enzymes like SOD and GSH-Px63.
Application of longer period of melatonin treatment, studying its effect on other organs and a modified dosage might result in greater benefits, may provide a new potential capability of melatonin.
It is concluded that the study confirmed that a dose of 40 mg kg–1 of GM resulted in hepatotoxicity as evidenced by significant increase in AST, ALT levels and the role of melatonin in ameliorating oxidative damage in liver tissue, by preventing the toxic effects of GM. Melatonin reduced MDA levels, hydroxyproline content and Protein carbonyl groups in liver and it increased GSH content and GST activity in liver.
Reports from animal studies have proved that melatonin is an ameliorating agent that works against nephrotoxicity and hepatotoxicity induced by GM, therefore, this study recommends application of melatonin treatment on humans, either by raising dietary intake of melatonin, or supplementation with supraphysiological dosages, to assessing clinical benefits and implications in humans, before general recommendation.
This study proves the potential effect of melatonin in prevention from hepatotoxicity induced by gentamicin. Patient could be advised to take melatonin while using gentamicin as a medication. This study will help the researchers to use natural antioxidant such as melatonin to strengthen the antioxidant defenses against reactive oxygen species, either separately or in combination with other natural products.
Author acknowledge Princess Nourah Bint Abdulrahman University, Kingdom of Saudi Arabia for providing facilities.