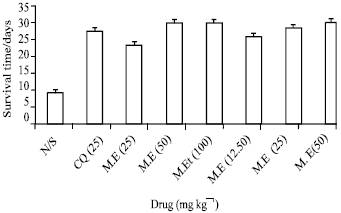
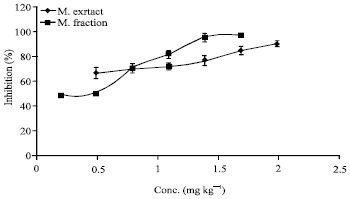
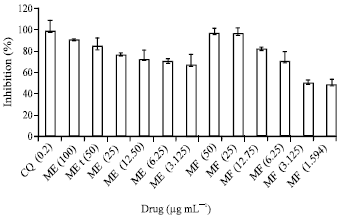
Research Article
The Potency of African Locust Bean Tree as Antimalarial
Department of Pharmacology and Therapeutics, College of Health Sciences, Bingham University, PMB 005, Karu, Nigeria
F. Tarfa
Department of Medicinal Chemistry and Quality Control, National Institute for Pharmaceutical Research and Development (NIPRD), PMB 21, Abuja, Nigeria
J.C. Aguiyi
Department of Pharmacology, Faculty of Pharmaceutical Sciences, University of Jos, PMB 2084, Jos, Nigeria