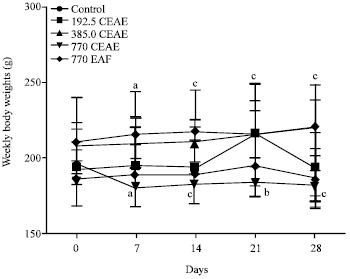
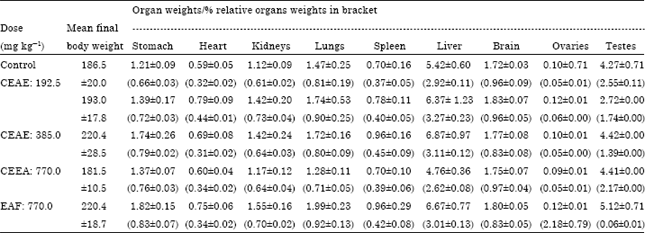
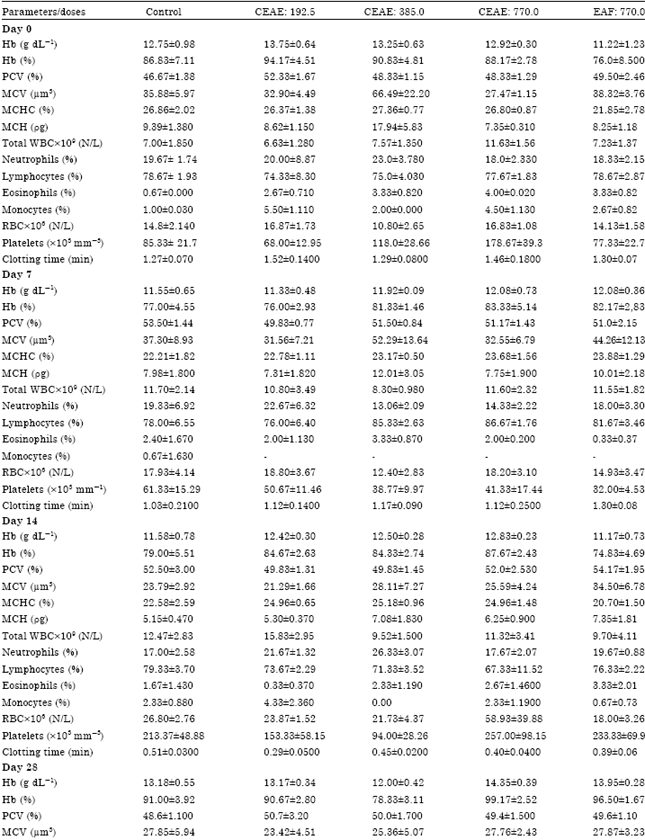
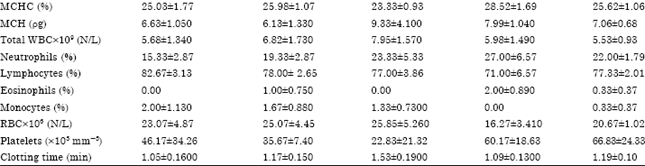
Research Article
Acute and Sub-acute Toxicity Profile of Carpolobia lutea Leaf Extract in Rats
Department of Pharmacology and Toxicology, Faculty of Pharmacy, Niger Delta University, Wilberforce Island, Bayelsa State, Nigeria
Adesite Samson Oluwaseyi
Department of Histopathology, University of Uyo Teaching Hospital, Uyo, Akwa Ibom State, Nigeria
Paul Alozie Nwafor
Department of Pharmacology and Toxicology, Faculty of Pharmacy, University of Uyo, Uyo, Akwa Ibom State, Nigeria