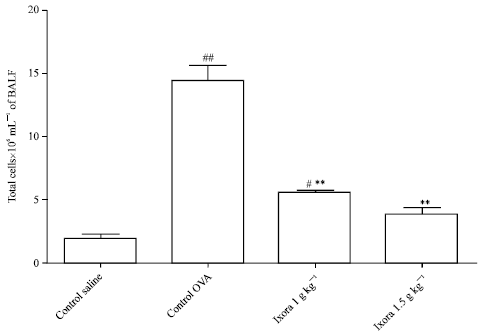
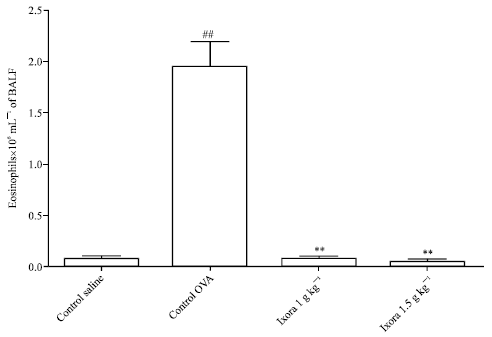
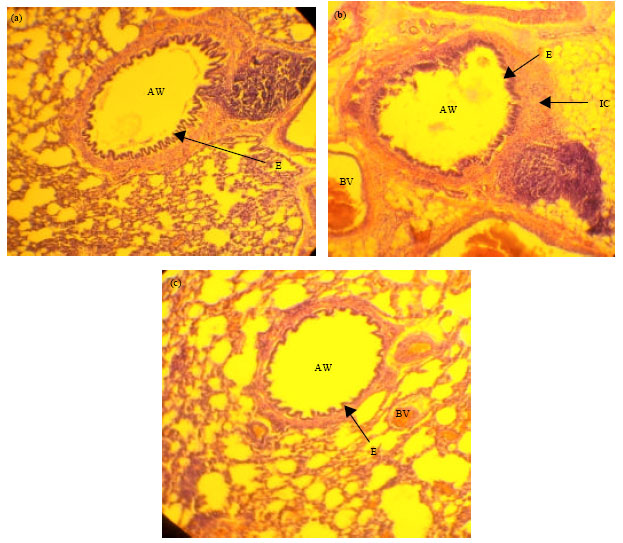
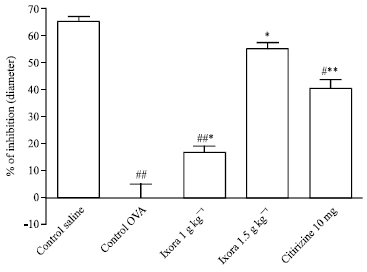
Research Article
Evaluation of Anti-asthmatic Activities of Ixora coccinea Linn (Rubiaceae)
Centre de Recherche et de Formation sur Les Plantes Medicinales (CERFOPLAM), Faculte des Sciences, Universite de Lome, BP: 1515, Lome, Togo
K. Metowogo
Centre de Recherche et de Formation sur Les Plantes Medicinales (CERFOPLAM), Faculte des Sciences, Universite de Lome, BP: 1515, Lome, Togo
A. Agbonon
Centre de Recherche et de Formation sur Les Plantes Medicinales (CERFOPLAM), Faculte des Sciences, Universite de Lome, BP: 1515, Lome, Togo
K. Eklu-Gadegbeku
Centre de Recherche et de Formation sur Les Plantes Medicinales (CERFOPLAM), Faculte des Sciences, Universite de Lome, BP: 1515, Lome, Togo
K. Aklikokou
Centre de Recherche et de Formation sur Les Plantes Medicinales (CERFOPLAM), Faculte des Sciences, Universite de Lome, BP: 1515, Lome, Togo
M. Gbeassor
Centre de Recherche et de Formation sur Les Plantes Medicinales (CERFOPLAM), Faculte des Sciences, Universite de Lome, BP: 1515, Lome, Togo