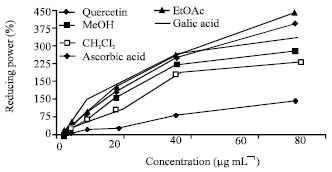
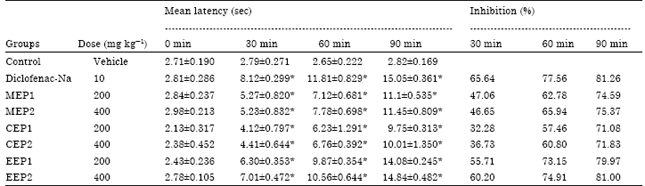
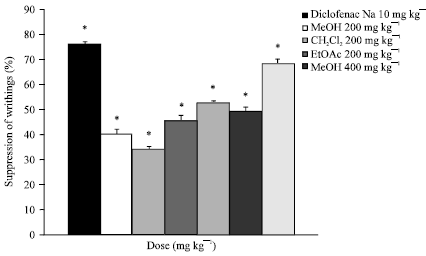
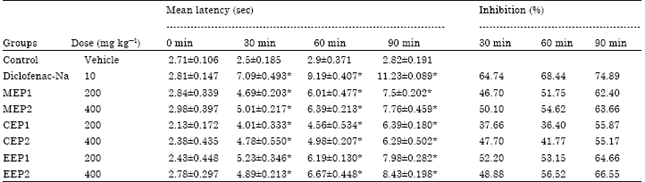
Research Article
Antioxidant, Analgesic and Anti-inflammatory Activities of the Herb Eclipta prostrata
Department of Pharmacy, Atish Dipankar University of Science and Technology, Dhaka, Bangladesh
M.B. Alam
Department of Pharmacy, Atish Dipankar University of Science and Technology, Dhaka, Bangladesh
N.S. Chowdhury
Department of Pharmacy, Manarat International University, Dhaka, Bangladesh
M. Asadujjaman
Department of Pharmacy, Atish Dipankar University of Science and Technology, Dhaka, Bangladesh
R. Zahan
Department of Pharmacy, Atish Dipankar University of Science and Technology, Dhaka, Bangladesh
M.M. Islam
Department of Pharmacy, Atish Dipankar University of Science and Technology, Dhaka, Bangladesh
M.E.H. Mazumder
Discipline of Biomedical Science, Faculty of Medicine, Cumberland Campus, University of Sydney, Australia
M.E. Haque
Discipline of Biomedical Science, Faculty of Medicine, Cumberland Campus, University of Sydney, Australia
A. Islam
Department of Pharmacy, Bangladesh University, Dhaka, Bangladesh