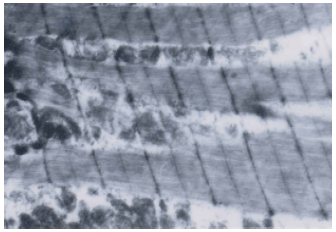
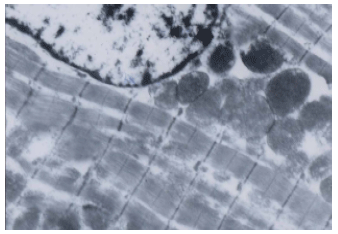
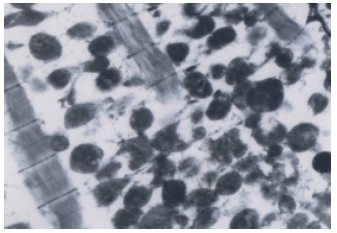
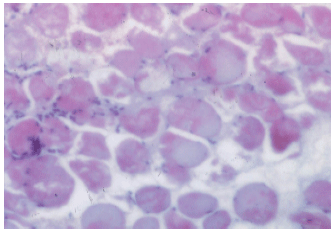
Research Article
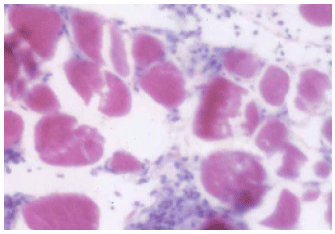
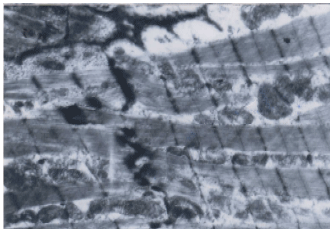
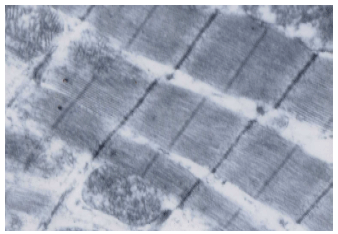
Coenzyme Q10 Ameliorates Statin-related Myotoxicity: A Biochemical and Histological Study
Forensic Medicine and Clinical Toxicology, Faculty of Medicine, Ain Shams University, Egypt
Ahmed Elsayed
Internal Medicine, National Research Centre, Egypt