ABSTRACT
The objective of this study was to mointer the metabolic disturbance in rats one hour after intoxication with one oral dose of ethanol. Rats orally administered with 10 g ethanol/kg body weight on 24 h empty stomach and sacrificed one hour later. Intoxicated rats recorded significant increase in blood alcohol level (alcohol dehydrogenase; ADH), aspartate (AST) and alanine (ALT) aminotransferase, lactate dehydrogenase (LDH) and gamma glutamyl transferase (GGT) enzyme activities. Carbohydrate deficient transferrin (CDT) level as a new marker for detection early independent ethanol intoxication recorded significant increase. Serum transferrin content was evaluated to avoid the false positive results of CDT. In conclusion, AST, ALT, LDH and GGT may be used as biomarkers for early diagnosis of ethanol toxicity. CDT plays a promise role for early diagnosis. CDT and the other biochemical tests especially GGT are recommended to be detected in parallel to improve the performance of either marker used alone.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jpt.2011.158.165
URL: https://scialert.net/abstract/?doi=jpt.2011.158.165
INTRODUCTION
Alcohol is not digested like other foods. It avoids the normal digestive process and goes directly to the blood stream (Levitt and Levitt, 1994). About 20% of the alcohol is absorbed directly into the blood through the stomach walls and 80% is absorbed into the bloodstream through the small intestine (Levitt et al., 1997). The maximum absorption rate is obtained with the consumption of alcohol solution on an empty stomach which also causes stomach ulceration (Karumi et al., 2008). The absorption rate may be less when alcohol is consumed with food (Horowitz et al., 1989).
Alcohol consumption leads to the production of the highly reactive ethanol metabolite, acetaldehyde, which may affect intestinal tight junctions, increase paracellular permeability and increase the solubility of penetrating chemicals (Fisher et al., 2010). This enhancement is beneficial if the chemicals involved are being used for therapeutic purposes, however exposure to potentially toxic chemicals which occurs both in the workplace and at home make increased absorption less desirable (Brand et al., 2006).
The clinical evaluation in the early phase of alcohol misuse is rarely useful for the diagnosis, since clinical signs are rather minimal (Sillanaukee, 1996). Furthermore, the common laboratory markers of alcohol misuse, such as serum gamma glutamyltransferase (GGT), aspartate aminotransferase (AST), alanine aminotransferase (ALT) and the erythrocyte Mean Cell Volume (MCV), have been used as a diagnostic tool (Sharpe et al., 1996) but with limited accuracy. Carbohydrate deficient transferrin, has high sensitivity in detecting persons with alcohol dependence and shows promise for identification of non-dependent hazardous drinking (0-70 g ethanol) (Schellenberg et al., 2005).
The aim of the present study was to evaluate the changes of some biochemical parameters 1 h after intoxication with one oral ethanol dose (10 g kg-1 b.wt.) that may used as biomarkers for detecting toxicity and for early diagnosis.
MATERIALS AND METHODS
Animals handling: Male Wistar albino rats (100: 120 g) were selected for this study. They were obtained from the Animal House, National Research Center, Egypt. All animals were kept in a control environment of air and temp with access of water and diet. Animals left free for a week for acclimatization. All animals were deprived of food for 24 h before the beginning of the experiment with excess of water. All procedures followed the ethical guidelines of the Ethical Committee of the Federal Legislation and National Institutes of Health Guidelines in USA approved by the Medical Ethical Committee of the National Research Centre in Egypt.
Ethanol intoxication: Ten grams ethanol/kg body weight (Brand et al., 2006) was orally given on 24 h empty stomach.
Experimental design: Thirty rats were divided into two groups. The first group (10 rats) served as healthy control group and orally administrated with 0.9 N physiological saline solution. The second group (20 rats) received the ethanol dose and sacrificed one hour after administration.
Sample preparations
Serum sample: Blood collected from each animal by puncture the sub-tongual vein in clean and dry test tube, left 10 min to clot and centrifuged at 3000 rpm for serum separation. The separated serum was stored at -80°C for further determinations.
Biochemical assays: Blood alcohol level was determined by the biochemical assay of alcohol dehydrogenase (ADH) (Vallee and Hoch, 1955). ADH catalyzes the oxidation of alcohol to acetaldehyde with the simultaneous reduction of nicotinamide adenine dinucleotide (NAD) to NADH. The consequent increase in absorbance at 340 nm is directly proportional to alcohol concentration in the sample.
Aspartate and alanine amintransferases were measured by the method of Gella et al. (1985), where the transfer of the amino group from aspartate or alanine, formed oxalacetate or pyruvate, respectively. The developed colour was measured at 520 nm.
Lactate dehydrogenase was assayed by the method of Babson and Babson (1973), where the reduction of NAD is coupled with the reduction of tetrazolium salt [2-(p-iodophenyl)-3-(p-nitrophenyl)-5-phenyl tetrazolium chloride] (INT). The resulted formazan of INT was measured colorimetrically at 503 nm.
Gamma-Glutamyl Transferase (GGT): The assay measures the cleavage of a specific GGT substrate (γ-glutamyl-p-ntiroanilide) by the enzyme to p-nitroaniline (pNA) product which is proportional to the level of GGT enzyme in the sample. The enzyme was measured at 405 nm (Szasz, 1969).
Serum CDT determinations were carried out using Kabi-Pharmacia kit (Sweden) by the method of Stibler et al. (1991). This test employs separation of transferrin isoforms on an anion exchange chromatography microcolumn followed by quantification with double antibody radioimmunoassay.
Transferrin level was estimated by the method of Matsumoto et al. (1991), where sandwich enzyme immunoassay of rat transferrin with two monoclonal antibodies was applied. Microtiter plates coated with one monoclonal antibody (15C2H3) were used and the captured transferrin was estimated with a horseradish peroxidase-conjugated Fab' fragment of another monoclonal antibody (22A06D2).
Statistical analysis: All data were expressed as mean±SD of rat numbers in each group. Statistical analysis was carried out using independent student t-test (Ronald et al., 1983).
Work performance: The present study was done on March-April, 2010 at Therapeutic Chemistry Department, National Research Center, Cairo, Egypt.
RESULTS AND DISCUSSION
In the present study, blood alcohol concentration (200 ±8.16 ng mL-1) in intoxicated rats recorded elevation by 53.84% as compared to the normal healthy rats (Fig. 1). This was in accordance with Brand et al. (2006) who observed a significant increase in blood alcohol level two hour after one oral dose of ethanol. Blood alcohol concentration was determined by measuring alcohol dehydrogenase activity, where in healthy rats the normal substrates are not the alcohols or aldehydes that react most readily with the enzyme but special substances such as vitamin A2 or farnesol (an intermediate in cholesterol biosynthesis) (Krebs and Perkins, 1970). Obviously, the toxic metabolic effects of ethanol oxidation are mainly due to increased liberation of ROS, production of deleterious active acetaldehyde, increased NADH/NAD ratio and disturbed intracellular calcium stores (Lieber, 2000; Soliman et al., 2006). Therefore, ethanol forms a toxic environment favorable to oxidative stress such as hypoxia, endotoxeamia and cytokines release (Bautisa and Spitzer, 1999).
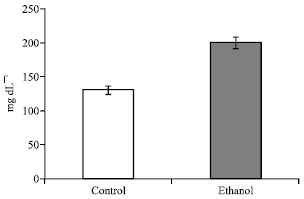 | |
| Fig. 1: | Effect of ethanol intoxication on blood alcohol concentration. Data are mean±SD of ten normal and twenty intoxicated rats. Blood alcohol value is expressed as mg dL-1. Significant value at p≤0.05; independent student t-test |
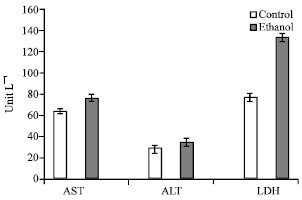 | |
| Fig. 2: | Effect of ethanol intoxication on serum AST, ALT and LDH. Data are mean± SD of ten normal and twenty intoxicated rats. Enzyme values are expressed as U L-1. Significant value at p≤0.05; independent student t-test |
Ethanol intoxicated rats recorded significant increase in AST (76.17±3.36 U L-1), ALT (33.96±3.12 U L-1) and LDH (133.29±3.66 U L-1) by 19.89, 22.24 and 16.03%, respectively (Fig. 2). In agreement with the present study Chen et al. (2003) observed a significant increase in AST and ALT after light/moderate drinkers (at least once per month; <210 g ethanol/week for men, <140 g ethanol/week for women). In addition, Onyesom and Anosike (2007) recorded elevation in AST and ALT in rabbits orally given 1.5 g ethanol kg-1 body weight as single daily dose for a continuous period of fifteen weeks. Moreover, Soliman et al. (2006) recorded a significant decrease in rat liver LDH after chronic ethanol toxicity. Although, AST, ALT and LDH are specific enzymes for detecting intoxication or monitoring liver diseases, they lack the specificity for alcohol toxicity (Chen et al., 2003). The increase in enzyme activities was mainly due to the effect of ethanol that interpolates and expands biomembranes leading to increase membrane fluidity and enzyme release (Yang et al., 2005; Soliman et al., 2006).
CDT is the newest procedure available to clinicians to assess harmful alcohol consumption and the first one to obtain approval from the US Food and Drug Administration (FDA) for identification of sustained alcohol consumption. In contrast, GGT, which is also FDA-approved, is intended to detect liver damage rather than alcohol misuse (Harasymiw and Bean, 2001). CDT (17.83±2.46 U L-1) and GGT (32.10±2.46 U L-1) recorded enhancement by 14.14 and 18.66%, respectively after intoxication with one oral dose of ethanol (Fig. 3). Present results were confirmed by many workers who postulated the use of CDT as a marker for heavily ethanol consumption (Harasymiw and Bean, 2001; Chen et al., 2003; Koch et al., 2004) neglecting to some extent its possible use as biomarker for independent ethanol toxicity and for early diagnosis. Van Pelt et al. (2000) documented the use of CDT and other biomarkers; AST, ALT and GGT in parallel to improve the performance of either marker used alone. They added that combination of CDT and GGT tests were more accuracy in detecting toxicity. In addition, Van Pelt and Azimi (1998) and Van Pelt et al. (2000) found that the amount of CDT will also be influenced by the rate of transferrin synthesis.
Transferrin is a plasma protein that carries iron through the bloodstream to the bone marrow, where red blood cells are manufactured, as well as to the liver and spleen. Structurally, transferrin is a polypeptide with two N-linked polysaccharide chains. These polysaccharide chains are branched with sialic acid (monosaccharide carbohydrate) residues (Sharpe, 2001). Various forms of transferrin exist, with differing levels of sialylation. The most common form is tetrasialotransferrin, with four sialic acid chains.
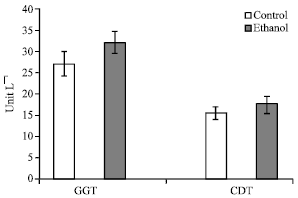 | |
| Fig. 3: | Effect of ethanol intoxication on serum GGT and CDT. Data of GGT are mean±SD of ten normal and twenty intoxicated rats. CDT values are mean±SD of eight normal and seventeen intoxicated rats. Enzyme values are expressed as U L-1. Significant value at p≤0.05; independent student t-test |
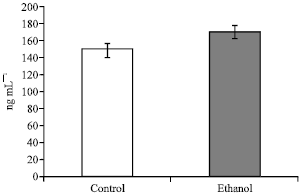 | |
| Fig. 4: | Effect of ethanol intoxication on serum transferrin level. Data are mean±SD of ten normal and twenty intoxicated rats. Values are expressed as ng mL-1. Significant value at p≤0.05; independent student t-test |
In persons who consume alcohol, the proportion of transferrin with zero, one, or two sialic acid chains is increased. These are referred to as carbohydrate-deficient transferrin (Sharpe, 2001).
In the present study, transferrin level (170.00 ± 8.11 ng mL-1) in intoxicated rats recorded significant increase by 13.33% (Fig. 4). This was in accordance with Golka et al. (2004) who observed elevation in ferritin and transferrin levels after ethanol intoxication. Van Pelt and Azimi (1998) and Bergstrom and Helander (2008) postulated a significant correlation between CDT and transferrin. Low iron status or high iron demand involves higher transferrin synthesis, probably with a proportional increase of CDT isoforms. However, CDT can induce false-positive outcome if the transferrin concentrations are low as in anemic state. In the present work, rats with low transferrin level <5 ng mL-1 (Matsumoto et al., 1991) were excluded to avoid false positive results of CDT. Control group under investigation represented 33.30% of the total animals. Rats with transferrin level <5 ng mL-1 were 6.66%. Intoxicated group represented 56.69% of the total animals and 10.01% of them had low transferrin level (<5 ng mL-1) (Fig. 5). Bergstrom and Helander (2008) recorded some other factors influenced CDT level as end stage liver disease, estrogens, pregnancy and inflammations. Wolff et al. (2010) added that hemodialysis patients showed high CDT that seems to be related to the low transferrin concentration associated with chronic kidney disease.
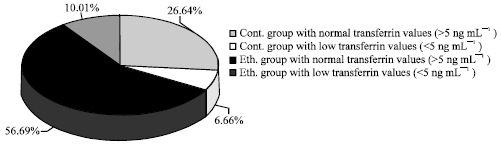 | |
| Fig. 5: | Percentages of animals with low and normal transferrin level. Rats with normal transferrin value were subjected to CDT test |
Present study was not concerned with these factors except transferrin level as we used healthy animals. All animals showed no signs of liver or kidney diseases or inflammations. Male animals let other factors like estrogen level and pregnancy are impossibly to be concerned.
CONCLUSIONS
One oral dose of ethanol intoxication leads to a disturbance of certain metabolic parameters that can be used as markers for early detection of toxicity. Combination of tests especially GGT and CDT is recommended for accuracy and specificity. CDT can be used as a marker for independent hazadours ethanol toxicity and is suitable for early diagnosis of toxicity. Certain diseases must be taken into considerations to be sure of CDT accuracy.
REFERENCES
- Babson, A.L. and S.R. Babson, 1973. Kinetic colorimetric measurement of serum lactate dehydrogenase activity. Clin. Chem., 19: 766-769.
CrossRefDirect Link - Brand, R.M., J.L. Jendrzejewski, E.M. Henery and A.R. Charron, 2006. A single oral dose of ethanol can alter transdermal absorption of topically applied chemicals in rats. Toxicol. Sci., 92: 349-355.
CrossRef - Chen, J., K.M. Conigrave, P. Macaskill, J.B. Whitfield and L. Irwig, 2003. Combining carbohydrate-deficient transferrin and gamma-glutamyltransferase to increase diagnostic accuracy for problem drinking. Alcohol. Alcoholism., 38: 574-582.
Direct Link - Fisher, S.J., P.W. Swaan and N.D. Eddington, 2010. The ethanol metabolite acetaldehyde increases paracellular drug permeability in vitro and oral bioavailability in vivo. J. Pharmacol. Exp. Ther., 332: 326-333.
Direct Link - Gella, F.J., T. Olivella, P.M. Cruz, J. Arenas, R. Moreno, R. Durban and J.A. Gomez, 1985. A simple procedure for routine determination of aspartate aminotransferase and alanine aminotransferase with pyridoxal phosphate. Clin. Chem. Acta, 153: 241-247.
PubMed - Harasymiw, J. and P. Bean, 2001. The combined use of the Early Detection of Alcohol Consumption (EDAC) test and carbohydrate-deficient transferrin to identify heavy drinking behaviour in males. Alcohol. Alcoholism, 36: 349-353.
PubMed - Horowitz, M., A. Maddox, M. Bochner, J. Wishart, R. Bratasiuk, P. Collins and D. Shearman, 1989. Relationships between gastric emptying of solid and caloric liquid meals and alcohol absorption. Am. J. Physiol. Gastrointest Liver Physiol., 257: G291-G298.
Direct Link - Krebs, H.A. and J.R. Perkins, 1970. The physiological role of liver alcohol dehydrogenase. Biochem. J., 118: 635-644.
Direct Link - Lieber, C.S., 2000. Hepatic metabolic and nutritional disorders of alcoholism: From pathogenesis to therapy. Critisism Rev. Clin. Lab. Sci., 37: 551-584.
Direct Link - Levitt, M.D. and D.G. Levitt, 1994. The critical role of the rate of ethanol absorption in the interpretation of studies purporting to demonstrate gastric metabolism of ethanol. J. Pharmacol., 269: 297-304.
Direct Link - Levitt, M.D., R. Li, E.G. Demaster, M. Elson, J. Furne and D.G. Levitt, 1997. Use of measurements of ethanol absorption from stomach and intestine to assess human ethanol metabolism. Am. J. Physiol. Gastrointest. Liver Physiol., 273: G951-G957.
Direct Link - Matsumoto, T., H. Shima, T. Tetsuya Kishi and T. Sato, 1991. Sandwich enzyme immunoassay for rat transferrin with two monoclonal antibodies and its application. Biochem. Med. Metab. Biol., 45: 188-196.
CrossRef - Schellenberg, F., R. Schwan, L. Mennetrey, M.N. Loiseaux, J.C. Pages and M. Reynaud, 2005. Dose-effect relation between daily ethanol intake in the range 0-70 grams and % cdt value: Validation of a cut-off value. Alcohol. Alcoholism, 40: 531-534.
CrossRef - Sharpe, P.C., 2001. Biochemical detection and monitoring of alcohol abuse and abstinence. Ann. Clin. Biochem., 38: 652-664.
Direct Link - Szasz, G., 1969. A kinetic photometric method for serum γ-glutamyl transpeptidase. Clin. Chem., 15: 124-136.
PubMed - Vallee, B. and F. Hoch, 1955. Yeast alcohol dehydrogenase, a zinc metalloenzyme. J. Am. Chem. Soc., 77: 821-822.
CrossRef - Van Pelt, J. and H. Azimi, 1998. False-positive CDTect values in patients with low ferritin values. Clin. Chem., 44: 2219-2220.
Direct Link - Van Pelt, J., G.L. Leusink, P.W.M. van Nierop and J.J. Keyzer, 2000. Test characteristics of carbohydrate-deficient transferrin and -glutamyltransferase in alcohol-using perimenopausal women. Alcoholism Clin. Exp. Res., 24: 176-179.
PubMed - Wolff, F., M. Mesquita, F. Corazza, A. Demulder and D. Willems, 2010. False positive carbohydrate-deficient transferrin results in chronic hemodialysis patients related to the analytical methodology. Clin. Biochem., 43: 1148-1151.
PubMed - Soliman, K.M., M.A. Hamed and S.A. Ali, 2006. Hepatoprotective effect of carnosine on liver biochemical parameters in chronic ethanol intoxicated rat. J. Medical Sci., 6: 528-536.
CrossRefDirect Link - Sillanaukee, P., 1996. Laboratory markers of alcohol abuse. Alcohol Alcoholism, 31: 613-616.
CrossRefPubMedDirect Link - Onyesom, I. and E.O. Anosike, 2007. Changes in Rabbit Liver function markers after chronic exposure to ethanol. Asian J. Biochem., 2: 337-342.
CrossRefDirect Link - Koch, H., G. Meerkerk, J.O.M. Zaat, M.F. Ham, R.J. Scholten and W.J. Assendelft, 2004. Accuracy of carbohydrate-deficient transferrin in the detection of excessive alcohol consumption: A systematic review. Alcohol Alcoholism, 39: 75-85.
PubMed - Karumi, Y., A.I. Augustine and I.A. Umar, 2008. Gastroprotective effects of aqueous extract of Adansonia digitata leaf on ethanol-induced ulceration in rats. J. Boil. Sci., 8: 225-228.
CrossRefDirect Link - Golka, K., R. Sondermann, S.E. Reich and A. Wiese, 2004. Carbohydrate-Deficient Transferrin (CDT) as a biomarker in persons suspected of alcohol abuse. Toxicol. Lett., 151: 235-241.
PubMed - Bergstrom, J.P. and A. Helander, 2008. HPLC evaluation of clinical and pharmacological factors reported to cause false-positive Carbohydrate-Deficient Transferrin (CDT) levels. Clin. Chem. Acta, 389: 164-166.
PubMed








