Research Article
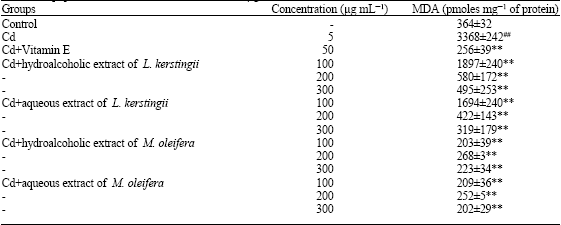
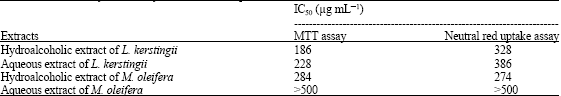
Protective Effect of Moringa oleifera Lam. and Lannea kerstingii Extracts Against Cadmium and Ethanol-induced Lipid Peroxidation
Department of Animal Physiology, Faculty of Sciences, University of Lome, Togo
K. Eklu-Gadegkeku
Department of Animal Physiology, Faculty of Sciences, University of Lome, Togo
T. Mobio
Department of Toxicology, Laboratory of Toxicology and Applied Hygiene, University Victor Segalen Bordeaux 2, Bordeaux, France
S. Moukha
Department of Toxicology, Laboratory of Toxicology and Applied Hygiene, University Victor Segalen Bordeaux 2, Bordeaux, France
A. Agbonon
Department of Animal Physiology, Faculty of Sciences, University of Lome, Togo
K. Aklikokou
Department of Animal Physiology, Faculty of Sciences, University of Lome, Togo
E.E. Creppy
Department of Toxicology, Laboratory of Toxicology and Applied Hygiene, University Victor Segalen Bordeaux 2, Bordeaux, France
M. Gbeassor
Department of Animal Physiology, Faculty of Sciences, University of Lome, Togo