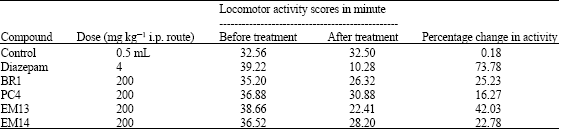
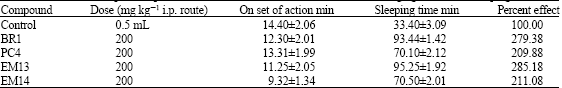Research Article
Anti Inflammatory, Antinociceptive and Central Nervous System Depressant Activities of Marine Bacterial Extracts
Centre for Research and Development, PRIST University, Vallam, Thanjavur, Tamil Nadu, India
S. Senthil Kumar
Laboratory for Conservation of Endangered Species, Centre for Cellular and Molecular Biology, Uppal Road, Hyderabad, Andhra Pradesh, India