Research Article
Effects of Mercury Exposure on Blood Chemistry and Liver Histopathology of Male Rats
Department of Zoology, College of Science, King Saud University, P.O. Box 2455, Riyadh-11451, Saudi Arabia
Mercury is a widespread environmental and industrial pollutant, which induces severe alterations in the tissues (Lund et al., 1993; Mahboob et al., 2001; Sener et al., 2007), causes numerous neurological abnormalities (Kingman et al., 2005; Auger et al., 2005) and produces peripheral neuropathy (Boyd et al., 2000; Chuu et al., 2007) in experimental animals and human beings.
Mercury poisoning can result from inhalation, ingestion, or absorption through the skin and may be highly toxic and corrosive once absorbed into blood stream. Furthermore, it combines with proteins in the plasma or enters the red blood cells but does not readily pass into the brain or fetus and instead, may enter other body organs (El-Shenawy and Hassan, 2008). The liver is a major site of metabolism for mercury and it can accumulate in the liver, resulting in severe hepatic damages. Previous studies have revealed that HgCl2 caused histopathological and ultrastructural lesions in the liver evidenced by periportal fatty degeneration and cell necrosis (El-Shenawy and Hassan, 2008). Schurz et al. (2000) stated that DNA was a vital molecule in the cell activities and was the main target for HgCl2-induced cell injuries.
In the present study, the toxic effects of mercury was evaluated by assessing toxicological end points in the form of body weight, blood chemistry and histopathology of liver in the male rats.
Animals
Twenty albino male rats Rattus norvegicus, weighing 150-170 g, aged 6-8 weeks, were randomly separated into two groups of 10 each. The first group formed the treated (with mercuric chloride) group and the second group was the control group. All animals were housed in standard metal cages in an air conditioned room at 22±3°C, 55±5% humidity and were provided with a standard laboratory diet and water ad libitum . They were obtained from animal house colony of the College of Pharmacy, King Saud University, Riyadh. All experimental procedures were conducted in accordance with the guide for the care and use of laboratory animals and in accordance with the Local Animal Care and Ethics Committee.
Treatment Design
Mercuric chloride (HgCl2) was obtained from Winlab Laboratory Chemicals, UK and was dissolved in sterilized distilled water giving a concentration of 20 ppm. This dose of HgCl2 was administered to the treated group of animals orally through their drinking water for eight weeks. The control group received sterilized distilled water only.
Experimental Assessments
Body Weight
Weight is a useful indicator of physical development. Thus the body weight of the animals was recorded at weekly interval until the end of the exposure period.
Blood Chemistry
At the end of the exposure period, the blood samples were collected from the retro-orbital plexus of all experimental animals under light anesthesia with ether (Sorg and Buckner, 1964). The blood was allowed to flow into sterilized centrifuge tubes and left to stand for 30 min before centrifugation to avoid hemolysis. The samples were centrifuged at 3000 g for 20 min. The clear supernatant was aspirated carefully for the estimation of various blood chemical parameters in the serum. The total blood chemistry parameters like alkaline phosphatase (ALP), alanine transaminase (ALT), aspartate transaminase (AST), gama-glutamyl transferase (GGT), Amylase (AMYL), cholesterol (CHOL), high density lipoproteins (HDLP); triglycerides (TG), creatinine (CREA), UREA, glucose (GLU) and potassium ion (K+) were assessed by the automatic analyzer apparatus (Reflotron plus, Roche, Germany) using commercially available Reflotron kits (Roche Diagnostics, Germany) for the above mentioned blood chemical parameters.
Histopathological Assessment
All rats were sacrificed after blood sampling by cervical dislocation. Their liver was dissected out and fixed in 10% Neutral Buffered Formalin (NBF) for 24 h and rinsed with 70% ethanol, dehydrated in serial dilutions of ethanol before embedding in paraffin wax. Paraffin blocks of the tissues were sectioned at 5-6 μm thickness in a rotary microtome. Interrupted serial sections of each block were processed for staining with haematoxyllin and eosin for histopathological details by the method described elsewhere (Bogdanovic et al., 2008). Photographs of the sections were taken at different magnifications in a Nikon Eclipse E600 Binocular Microscope, fitted with Nikon Digital Camera model DXM1200F, Japan.
Statistical Analysis
Data of the two groups of animals were analyzed by Student-Newman-Keuls multiple comparison test of ANOVA and p<0.05 was considered as significant.
Body Weight
The present results clearly showed that the body weight of the HgCl2 treated rats lagged behind their controls significantly (p<0.05) from the first week after exposure (Fig. 1), however, the decline in the body weight gain of the treated animals became highly significant (p<0.005) from 3rd week onwards until 8th week of HgCl2 exposure as compared to their controls (Fig. 1).
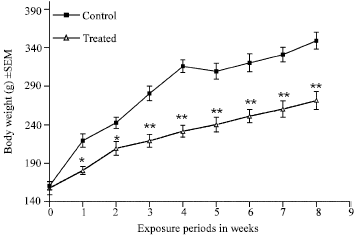 | |
| Fig. 1: | Effect of mercury exposure (20 ppm) through drinking water, on the profile of body weight gain of male rats for eight weeks. * and ** indicate statistically significant (p<0.05 and p<0.005, respectively) as compared to the control by ANOVA. |
| Table 1: | Blood chemical indexes in the serum of male rats exposed to mercuric chloride (20ppm) in their drinking water for eight weeks |
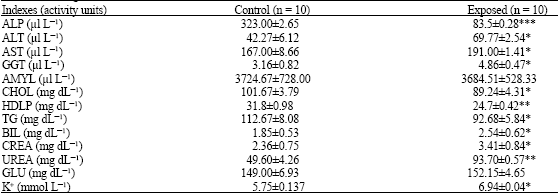 | |
| Values are expressed as the Means±SEM. *p<0.05, **p<0.01 and ***p<0.001 as compared to the controls evaluated by Student-Newman-Keuls multiple comparison test of ANOVA. n: No. of animals, ALP: alkaline phosphatase, ALT: Aalanine transaminase, AST: Aaspartate transaminae, GGT: Gama-glutamyl transferase, AMYL: Amylase, CHOL: Cholesterol, HDLP: High density lipoprotein; TG: Triglycerides, BIL: Bilirubin, CREA: Creatinine, UREA: Urea, GLU: Glucose, K+: Potassium ion | |
Blood Chemistry
Among the observed parameters of blood chemistry, ALP decreased very significantly (p<0.001) in the HgCl2 exposed animals, whereas the significance of CHOL, HDLP and TG decrease was comparatively low (p<0.05) in the treated animals (Table 1). On the other hand, ALT, AST, GGT, BIL, CREA, UREA and K+ were significantly (p<0.05) increased in the HgCl2 exposed animals. However, AMYL and GLU levels remained unaffected by HgCl2 exposure as compared to their control (Table 1).
Histopathological Observations
At light microscopical level the liver of the control animals is found to be composed of a continuous compact field of hepatocytes interspersed with blood sinusoids and intermittent islands of connective tissues enclosing the bile ducts, venous and arterial vessels (Fig. 2).
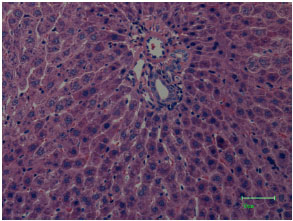 | |
| Fig. 2: | Light microscopical observations on the histological structures of the liver of normal untreated control male rats (staining by eosin and haematoxylin; magnified 100x), showing intact basic liver structures like central vein, bile duct, arterial vessel and hepatocytes. Magnification bar = 50 μm |
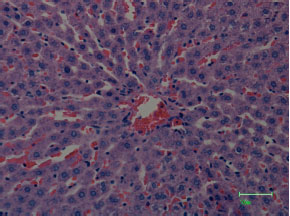 | |
| Fig. 3: | Light microscopical observations on the qualitative and structural damages of the liver tissue of male rats exposed to mercury (20 ppm) for eight weeks, showing damaged central vein, deformed and degenerated hepatocytes and necrosis of tissues (staining by eosin and haematoxylin; magnified 100x). Magnification bar = 50 μm |
The hepatocytes are radially organized in hepatic lobules, which in most cases are very tortuous and branched, with a large central vein. Close to the blood sinusoids, other structures are located like endothelial cells, bile canaliculi and reticular fibers (Fig. 2). After HgCl2 treatment, the liver tissue was completely damaged and qualitative degenerative and necrotic changes were observed in almost all structures of the liver tissue (Fig. 3).
In the present study, decrease in body weight gain, alteration in the normal levels of various blood chemical parameters accompanied by the histopathological necrosis and degenerative changes in the liver tissue were the main toxic effects observed in the male rats exposed orally to HgCl2 for eight weeks. Such decrease in body weight gain of rats due to mercury exposure has been reported earlier also (Jadhav et al., 2007). Furthermore, it has also been reported that mercury exposure increased the activities of AST, urea and creatinine, whereas glucose level remained unaffected and qualitative vascular degenerative and necrotic changes were also observed in the liver of male rats (Jadhav et al., 2007). These observations are clearly in agreement with the present findings.
Elevation in the activity of serum ALT, a liver cytoplasmic enzyme, indicates for necrotic lesions in the liver, while a decrease in serum ALP level indicates for no congestion or cholestasis (El-Demerdash, 2001; Reus et al., 2003; Sharma et al., 2002). These researchers (loc cit) reported that mice treated with HgCl2 showed significant elevations in serum glutamate oxaloacetate transaminase (SGOT) and serum glutamate pyruvate transaminase (SGPT) activities, whereas a significant decline in the ALP activity. The present study also shows that rats exposed to HgCl2 have elevated levels of serum ALT and AST, whereas a reduced level of ALP as compared to the saline control group, indicating clearly that our results are in agreement with other studies (El-Demerdash, 2001; Reus et al., 2003; Sharma et al., 2002). Furthermore, Jagadeesan and Pillai (2007) also reported significant increase in the level of serum AST and ALT in rats due to HgCl2 treatment for longer time period (30 days). Recently, in another study, besides a significant rise in the serum ALT and AST, a significant rise in GGT, BIL and CREA has also been reported in mercury exposed rats (Singh et al., 2007). On the other hand, in non-rodent model also (teleost fish), Sastry and Sharma (1980) reported that ALP activity decreased in acute exposure to HgCl2 and increased in chronic exposure to HgCl2, however, there was elevation of both SGPT and SGOT either in acute or chronic exposure to HgCl2. Thus, ALT activity in serum, could serve as a marker enzyme to evaluate functional status of liver as suggested by Sobutskii et al. (2007).
Altogether, the present results in the light of the above cited literature clearly indicate that increase in serum ALT and AST and decrease in serum ALP, can be used as potential enzyme biomarkers for mercury-induced hepatotoxicosis which ultimately affects the general health by altering the functional and structural integrity of liver. The other blood parameters like GGT, BIL and CREA may also serve as possible bio-indicators for mercury poisoning. However, in order to establish these serum enzyme levels as biomarkers for mercury poisoning, further detailed studies are required at experimental as well as clinical levels.