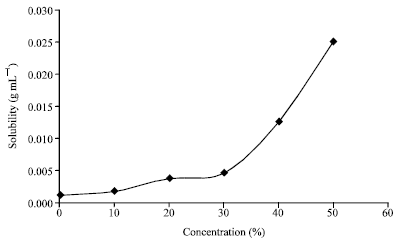
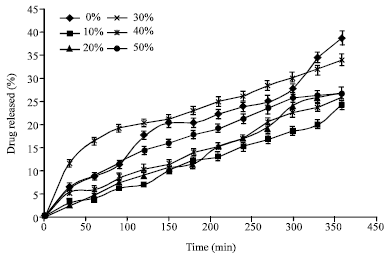
Research Article
Hydro-Alcoholic Media: An Emerging in vitro Tool for Predicting Dose Dumping from Controlled Release Matrices
Department of Pharmaceutical Technology and Raw Material Development, National Institute for Pharmaceutical Research and Dev elopment Iclu, P.M.B. 21, Garki-Abuja, Nigeria
P.I. Nwabunike
Department of Pharmaceutical Technology and Raw Material Development, National Institute for Pharmaceutical Research and Dev elopment Iclu, P.M.B. 21, Garki-Abuja, Nigeria
C.Y. Isimi
Department of Pharmaceutical Technology and Raw Material Development, National Institute for Pharmaceutical Research and Dev elopment Iclu, P.M.B. 21, Garki-Abuja, Nigeria
O.O. Kunle
Department of Pharmaceutical Technology and Raw Material Development, National Institute for Pharmaceutical Research and Dev elopment Iclu, P.M.B. 21, Garki-Abuja, Nigeria
S.I. Ofoefule
Department of Pharmaceutical Technology and Industrial Pharmacy, University of Nigeria, Nsukka, Enugu State, Nigena