Research Article
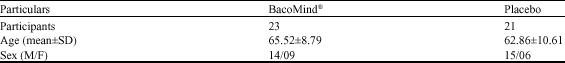
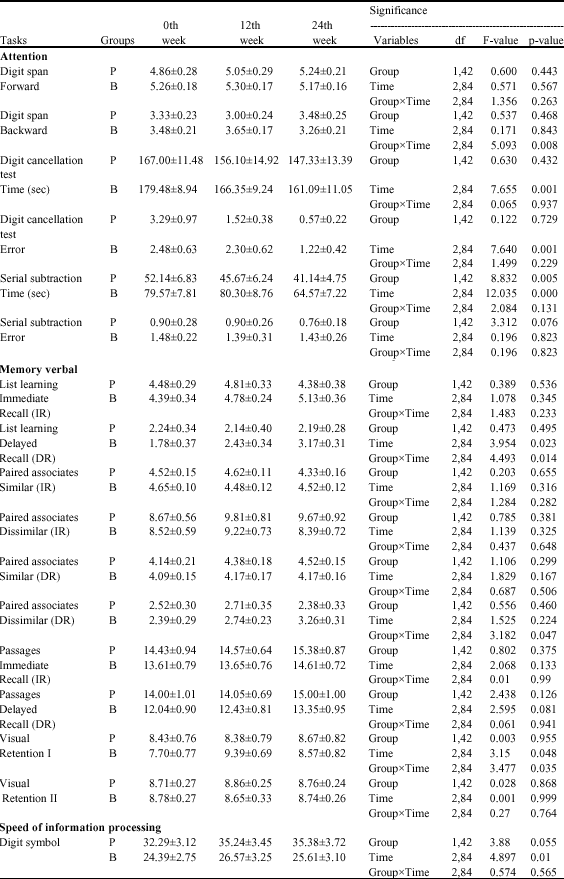
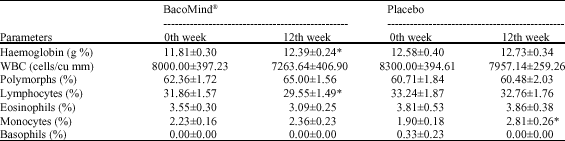
Efficacy and Tolerability of BacoMind®on Memory Improvement in Elderly Participants - A Double Blind Placebo Controlled Study
Consultant Physician, Ahmedabad, India
Rajeshwari P. Desai
Consultant Clinical Psychologist, Ahmedabad, India
Vinod S. Saxena
R and D Centre, Natural Remedies Pvt. Ltd., Bangalore-560100, Karnataka, India
K. Pravina
R and D Centre, Natural Remedies Pvt. Ltd., Bangalore-560100, Karnataka, India
P. Wasim
R and D Centre, Natural Remedies Pvt. Ltd., Bangalore-560100, Karnataka, India
P. Geetharani
R and D Centre, Natural Remedies Pvt. Ltd., Bangalore-560100, Karnataka, India
J. Joshua Allan
R and D Centre, Natural Remedies Pvt. Ltd., Bangalore-560100, Karnataka, India
K. Venkateshwarlu
R and D Centre, Natural Remedies Pvt. Ltd., Bangalore-560100, Karnataka, India
A. Amit
R and D Centre, Natural Remedies Pvt. Ltd., Bangalore-560100, Karnataka, India