Research Article
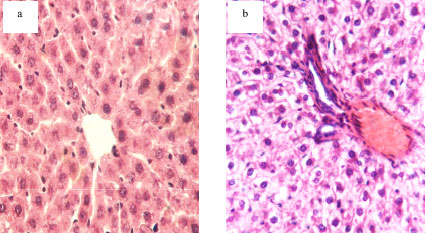
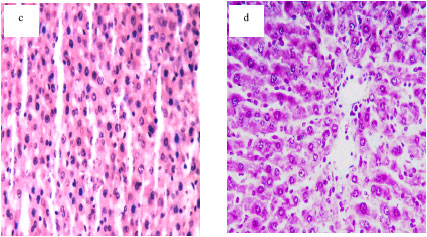
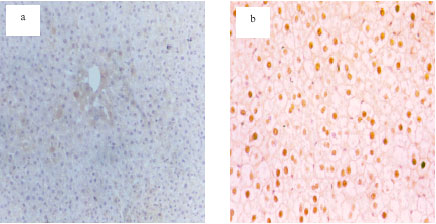
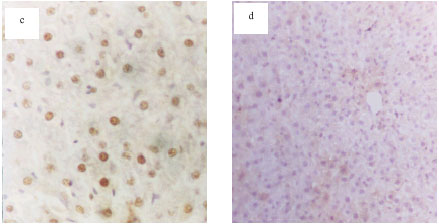
Protective Effects of Propolis Against the Amitraz Hepatotoxicity in Mice
Department of Zoology, Faculty of Science, Fayoum University, Egypt
Ayman Ahmed Owayss
Depatment of Protection Plant, Faculty of Agriculture, Fayoum University, Egypt