Research Article
Molecular Modelling Analysis of the Metabolism of Ramelteon
Discipline of Biomedical Science, School ofMedical Sciences, Faculty of Medicine, Cumberland Campus, C42 The University of Sydney, Lidcombe, NSW, Australia
Ramelteon [(S)-N-[2-(1,6,7,8-tetrahydro-2H-indeno-(5,4-b]furan-8-yl) ethyl]propanamide; TAK-375; RMT] is an agonist of the melatonin receptor, used for treatment of insomnia (Kato et al., 2005; Karim 2006; Borja and Daniel, 2006; Nguyen et al., 2005). The drug can exist in both R- and S-enantiomers. However, the marketed drug is found to be in the S-form. RMT has a high selectivity for the MT1 and MT2 subtype melatonin receptors that have been implicated in the maintenance of circadian rhythms, over MT3 receptor that is responsible for other melatonin functions. By incorporating methoxy moiety of melatonin into a conformationally constrained indeno [5,5-b] furan, the oxygen is locked into an orientation favourable for optimal and selective binding to the MT1 receptor (Hegde and Schmidt, 2006). The drug also lacks affinity for GABA (γ-aminobutyric acid) receptor complex and neurotransmitter, dopaminerigic, opiate and benzodiazepine receptors, suggesting an improved safety devoid of the abuse potential of the hypnotic drugs that target these receptors. The recommended dose of RMT is 8 mg taken within 30 min of going to bed. Clinically important sedative and performance-impairing effects of the drug are found to be minimal even at doses as high as 64 mg (Karim et al., 2006).
RMT is readily soluble in organic solvents such as methanol, ethanol and dimethyl sulfoxide and very slightly soluble in water and aqueous buffers from pH 3 to 11. It is rapidly absorbed reaching peak serum concentrations within one hour (Stubbs and Karim, 2003). RMT fulfils the two important pharmacokinetic characteristics of a sleep-promoting agent namely fast absorption (for the rapid onset of pharmacologic effect) and short half-life to minimize the next-day effects. Of the administered RMT, 84% is excreted in the urine and 4% in the faeces, indicating high absorption of the drug. Excretion of unchanged drug in urine is found to be negligible, suggesting extensive first-pass metabolism. RMT is also rapidly eliminated with a mean elimination half-life of 1.36 h after an 8 mg dose. RMT is metabolized primarily via oxidation to hydroxyl and carbonyl derivatives with secondary metabolites resulting from glucuronidation (Karim, 2006). CYP1A2 is the major isoenzyme involved in the hepatic metabolism of RMT (Hegde and Schmidt, 2006). CYP2C and CYP3A4 are also involved in the metabolism of the drug. The metabolite 1 (termed RMTM1) is formed by opening of the heterocyclic ring; it has a carboxyl and a hydroxyl group. The metabolite 2 (denoted as RMTM2) is formed by aliphatic hydroxylation and is the principal metabolite found in serum. The metabolite 3 (termed RMTM3) is formed from oxidation of five-membered alicyclic ring of RMT. The metabolite 4 (denoted as RMTM4) is the corresponding oxidation product of RMTM2 (Fig. 1).
The role of metabolites in the pharmacology of RMT is not clearly understood (Karim, 2006). The pharmacologic activity of RMTM1 and RMTM2 are found to be low as are their serum concentrations. RMTM4 also has negligible activity. The most active metabolite is RMTM2 although its activity is 30-fold lower than that of the parent drug. However, the serum levels of RMTM2 are found to exceed those of the parent drug by approximately 40-fold (Karim et al., 2006), indicating that the metabolism of RMT takes place rapidly.
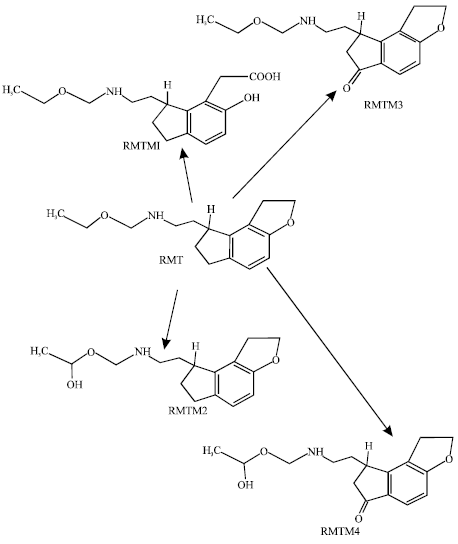 | |
| Fig. 1: | Metabolic pathways for RMT based on Karim (Karim, 2006) |
The adverse effects of RMT (observed in 5% of patients) include somnolence, dizziness, nausea, fatigue, headache and insomnia (Hegde and Schmidth, 2006). The drug should not be used in patients with hepatic impairment and precaution should be exercised in its co-administration with CYP inhibitors. In this study, molecular modelling analyses have been carried out using the program Spartan 02 (Spartan, 2002) to investigate the relative stability of RMT and its metabolites with the aim of providing a better understanding of their relative toxicity. The study was carried out in the Discipline of Biomedical Science, Faculty of Medicine, The University of Sydney during January to March 2007. To the best of my knowledge, no such study on ramelteon was done previously.
COMPUTATIONAL METHODS
The geometries of RMT and its metabolites RMTM1, RMTM2. RMTM3 and RMTM4 have been optimised based on molecular mechanics, semi-empirical and DFT calculations, using the molecular modelling program Spartan 02. Molecular mechanics calculations were carried out using MMFF force field. Semi-empirical calculations were carried out using the routine PM3. Density Functional Theory (DFT) calculations were carried at B3LYP/6-31G* level. In optimization calculations, a RMS gradient of 0.001 was set as the terminating condition. For the optimised structures, single point calculations were carried out to give heat of formation, enthalpy, entropy, free energy, dipole moment, solvation energy, energies for Highest Occupied Molecular Orbital (HOMO) and Unoccupied Molecular Orbital (LUMO). The order of calculations: molecular mechanics followed by semi-empirical followed by DFT ensured that the structure was not embedded in a local minimum. To further check whether the global minimum was reached, some calculations were carried out with improvable structures. It was found that when the stated order was followed, structure corresponding to the global minimum or close to that could ultimately be reached in all cases. Although RMS gradient of 0.001 may not be sufficiently low for vibrational analysis, it is believed to be sufficient for calculations associated with electronic energy levels.
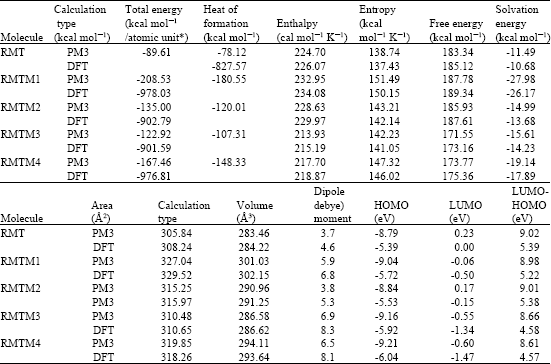
Table 1 shows the total energy, heat of formation as per PM3 calculation, enthalpy, entropy, free energy, surface area, volume, dipole moment and energies of HOMO and LUMO as per both PM3 and DFT calculations for RMT and its metabolites RMTM1, RMTM2. RMTM3 and RMTM4. Figure 2-6 give the regions of negative electrostatic potential (greyish-white envelopes) in (a), HOMOs (where red indicates HOMOs with high electron density) in (b), LUMOs in (c) and density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) in (d) as applied to the optimised structures of RMT and its metabolites RMTM1, RMTM2. RMTM3 and RMTM4.
All the metabolites of RMT are found to have higher solvation energies than the parent drug, indicating that the metabolites would have a greater solubility in water and hence more easily excreted in the urine. It was noted earlier that the serum levels of RMTM2 exceed those of the parent drug by approximately 40-fold (Karim et al., 2006), that can be explained in terms of rapid metabolism of the drug.
The LUMO-HOMO energy differences for RMT and its metabolites RMTM1, RMTM2, RMTM3 and RMTM4 from DFT calculations are found to range from 4.6 to 5.4 eV, indicating that the compounds would be moderate to highly inert kinetically.
In the case of RMT, RMTM1, RMTM2, RMTM3 and RMTM4, the electrostatic potential is found to be more negative around the various oxygen centers, indicating that the positions may be subject to electrophilic attack.
| Table 1: | Calculated thermodynamic and other parameters of RMT and its metabolites RMTM1, RMTM2, RMTM3 and RMTM4 |
 | |
| *: In atomic units from DFT calculation | |
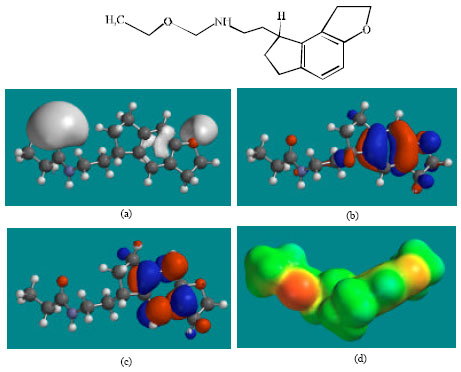 | |
| Fig. 2: | Structure of RMT giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
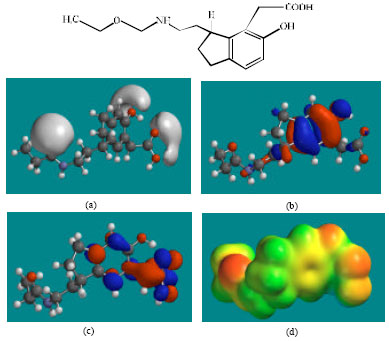 | |
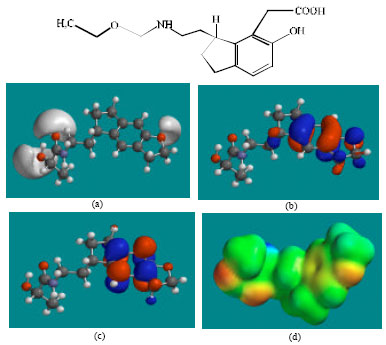
| Fig. 3: | Structure of RMTM1 giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
 | |
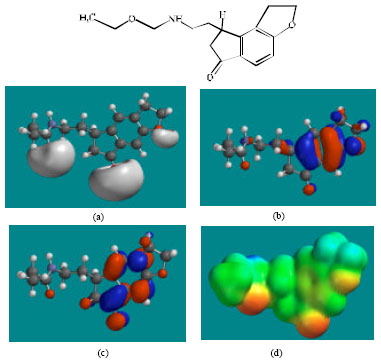
| Fig. 4: | Structure of RMTM2 giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
 | |
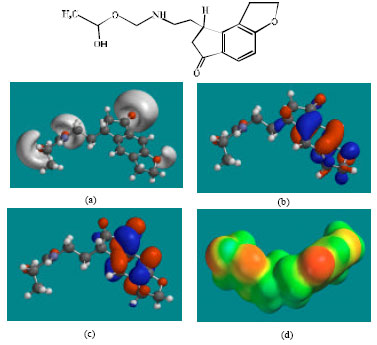
| Fig. 5: | Structure of RMTM3 giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
 | |
| Fig. 6: | Structure of RMTM4 giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
In the case of RMT, RMTM2, RMTM3 and RMTM4, both the HOMOs with high electron density and the LUMOs are found to be close to mainly the non-hydrogen atoms of the aromatic ring. In the case of RMTM1 also, the HUMOs with high electron density are centred mainly on the non-hydrogen atoms of the aromatic ring whereas the LUMOs are found to have a greater spread.
The overlap of HOMO with high electron density and region of negative electrostatic potential at some positions, gives further support to the idea that the positions may be subject to electrophilic attack.
The molecular surfaces of RMT, RMTM2, RMTM3 and RMTM4 are found to abound in neutral (green) and negative (yellow and red), neutral (green) regions so that the compound may be subject to lyophilic and electrophilic attacks. The presence of electron-rich regions on the molecular surface is most widespread in the case of RMTM1 than RMT or any other metabolite so that the compound may be most subject to electrophilic attacks.
When the surface area and volume of RMT are compared with those of its metabolites, it is found that the values of RMTM2 and RMTM3 are similar to those of the parent drug (Table 1). Although the values of RMTM3 are more similar than those of RMTM2, RMTM2 is found to be more active than RMTM3. The results indicate that the incorporation of ring carbonyl group in RMTM3 than the presence of OH group in the open chain of RMTM2 is a greater determinant of loss in activity.
Ramelteon (RMT) is an agonist of the melatonin receptor, used for treatment of insomnia. The drug has a high selectivity for the MT1 and MT2 subtype melatonin receptors that have been implicated in the maintenance of circadian rhythms, over MT3 receptor that is responsible for other melatonin functions. Molecular modelling analyses based on semi-empirical and DFT calculations show that RMT and its metabolites have moderately large to large LUMO-HOMO energy differences so that the compounds would be moderate to highly inert kinetically. The molecular surfaces of RMT and its metabolites are found to abound in neutral (green) and electron-rich (red and yellow) regions so that the compounds may be subject to both lyophilic and nucleophilic attacks. The absence of any significant amount of electron-deficient regions on the molecular surface indicates that RMT and its metabolites may not react with cellular nucleophiles such as glutathione and nucleobases in DNA so that the compounds may not induce cellular toxicity (that results from glutathione depletion) and may not also cause DNA damage (associated with oxidation of nucleobases in DNA). Rather, RMT and its metabolites may behave more like antioxidants.
Fazlul Huq is grateful to the Discipline of Biomedical Science, School of Medical Sciences, The University of Sydney for the time release from teaching.