Research Article
Molecular Modelling Analysis of the Metabolism of Mefenamic Acid
School of Biomedical Sciences, Faculty of Health Sciences, The University of Sydney, P.O. Box 170, Lidcombe, NSW 1825, Australia
Non-steroidal anti-inflammatory drugs (NSAIDs) are used in the long term treatment of rheumatoid arthritis and osteoarthritis to relieve pain and inflammation (Brooks, 1998). NSAIDs constitute a heterogeneous family of drugs but many have a common carboxylic acid moiety. An example of NSAID containing carboxylic moiety is mefenamic acid (MFA). Although MFA is widely used in analgesia (Siraki et al., 2005), its use has been implicated in several cases of nephrotoxicity including acute renal failure and tubulointestinal nephritis. NSAIDs produce a broad range of toxic side effects, frequently causing gastrointestinal (GI) toxicity that can result in ulceration, bleeding and perforation (Murray and Brater, 1993; James and Hawkey, 2003). The toxicity of NSAIDs is mainly attributed to (1) the induction of prostaglandin synthase COX-1 (Cyclooxygenase 1) activity that inhibits production of prostaglandin in the GI tract resulting in accumulation of intracellular arachidonic acid, (2) the induction of mitochondrial injury in hepatocytes through uncoupling of oxidative phosphorylation (Moreno-Sanchez et al., 1999; Mahmud et al., 1996) and (4) the production of reactive metabolites that bind covalently to critical cellular proteins (Mahmud et al., 1996; Hubert et al., 2005). For NSAIDs containing benzoic acid moiety, cytotoxicity is also correlated parabolically with lipophilicity.
In humans MFA is metabolized by both phase I and phase II enzymes. A major biotransformation and elimination pathway is conjugation with glucuronic acid at the carboxyl site catalysed by the uridine diphosphoglucuronosyl transferase (UGT) superfamily of enzymes (Graebing et al., 2004; Pritchard et al., 1994). Acyl glucuronides of MFA have been demonstrated to covalently bind to intra- and extracellular proteins. However, the toxicological consequences of this binding remain unclear (Boelsteri et al., 1995; King and Dickinson, 1993). Three MFA glucuronides were identified and isolated from human urine (McGurk et al., 1996). Although acyl glucuronides are unstable and can undergo acyl migration and hydrolysis, it has been found that inhibition of acyl glucuronidation results in increased NSAID cytotoxicity (Siraki et al., 2005). Thus it has been suggested that MFA acyl-glucuronides are less toxic. Two oxidative metabolites of MFA namely 3’-hydroxymethylmefenamic acid (3’HMMFA) and 3’-carboxymefenamic acid (3’CMFA) have also been identified. Cytochrome P450 enzymes CYP2C9 and CYP2C19 appear to be involved in the oxidative metabolism of MFA. 3’HMMFA and 3’CMFA can undergo acyl glucuronidation to produce 3’-hydroxymethylmefenamic acid glucuronide (3’HMMFA-Glu) and 3’-carboxymefenamic acid glucuronide (3’CMFA-Glu), respectively (Fig. 1).
In this study, molecular modelling analyses have been carried out using the programs HyperChem 7.0 (HyperChem, 2002) and Spartan ’02 (Spartan, 2002) to investigate the relative stability of MFA and its metabolites with the aim of providing a better understanding on the relative toxicity due to MFA and its metabolites.
Computational Methods
The geometries of MFA, MFA-Glu, 3’HMMFA, 3’CMFA, 3’HMMFA-Glu and 3’CMFA-Glu have been optimised based on molecular mechanics, semi-empirical and DFT calculations, using the molecular modelling programs Spartan ’02 and HyperChem 7.0. Molecular mechanics calculations were carried out using MM+ force field. Semi-empirical calculations were carried out using the routine PM3. DFT calculations were carried using the program Spartan ’02 at B3LYP/6-31G* level. For the optimised structures, single point calculations were carried to give heat of formation, enthalpy, entropy, free energy, dipole moment, solvation energy, energies for HOMO and LUMO. The order of calculations: molecular mechanics followed by semi-empirical followed by DFT minimized the chances of the structures being trapped in local minima rather reaching global minima. To further check whether the global minimum was reached, some calculations were carried out with improvable structures. It was found that when the stated order was followed, structures corresponding to global minimum or close to that were reached in most cases. Although RMS gradient of 0.001 may not be sufficiently small for vibrational analysis, it is believed to be sufficiently low for calculations associated with electronic energy levels. For the optimised structures, single point calculations were carried to give heat of formation, enthalpy, entropy, free energy, dipole moment and solvation energy, HOMO and LUMO.
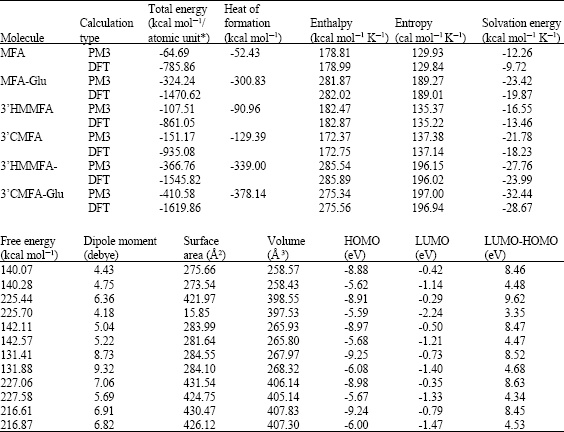
Table 1 gives the total energy, heat of formation as per PM3 calculation, enthalpy, entropy, free energy, dipole moment, energies of HOMO and LUMO as per both PM3 and DFT calculations for MFA and its metabolites MFA-Glu, 3’HMMFA, 3’CMFA, 3’HMMFA-Glu and 3’CMFA-Glu. Figure 2-7 give the regions of negative electrostatic potential (greyish-white envelopes) in (a), HOMOs (where red indicates HOMOs with high electron density) in (b), LUMOs in (c) and surface charges (where red indicates negative, blue indicates positive and green indicates neutral) in (d) as applied to the optimised structures of MFA and its metabolites MFA-Glu, 3’HMMFA, 3’CMFA, 3’HMMFA-Glu and 3’CMFA-Glu.
The calculated solvation energies of MFA and its metabolites MFA-Glu, 3’HMMFA, 3’CMFA, 3’HMMFA-Glu and 3’CMFA-Glu from PM3 calculations in kcal mol-1 are -12.26, -23.42, -16.55, -21.78, -27.76 and -32.44, respectively and their dipole moments from DFT calculations are 4.75, 4.18, 5.22, 9.32, 5.69 and 6.82, respectively. It can be seen that all the metabolites of MFA have greater solvation energy than the parent drug, indicating that they would be more soluble in water. The increase in solubility in water is to be expected since hydroxylation, demethylation and glucuronidation all effectively serve to increase polarity of the molecules.
| Table 1: | Calculated thermodynamic and other parameters of MFA and its metabolites |
 | |
| *In atomic units from DFT calculations | |
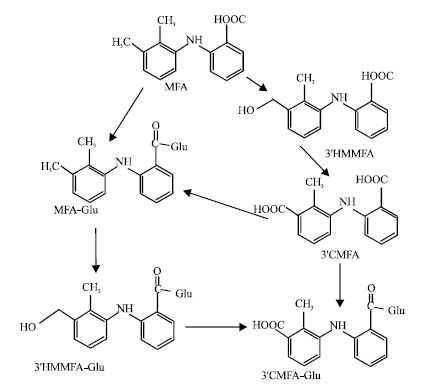 | |
| Fig. 1: | Proposed metabolic pathways for niclosamide and its metabolites (Graebing et al., 2004) |
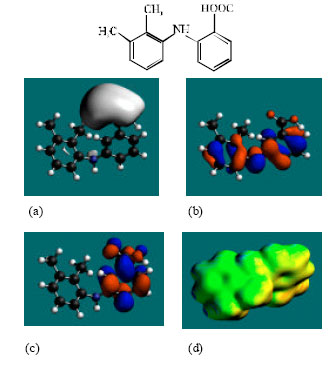 | |
| Fig. 2: | Structure of MFA giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) surface electric charges (where red indicates negative, blue indicates positive and green indicates neutral) |
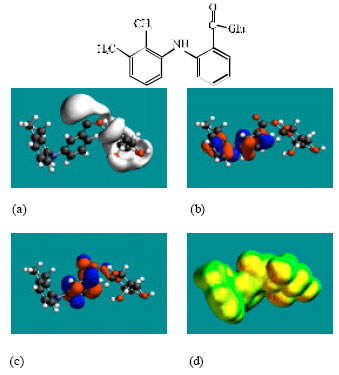 | |
| Fig. 3: | Structure of MFA-Glu giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) surface electric charges (where red indicates negative, blue indicates positive and green indicates neutral) |
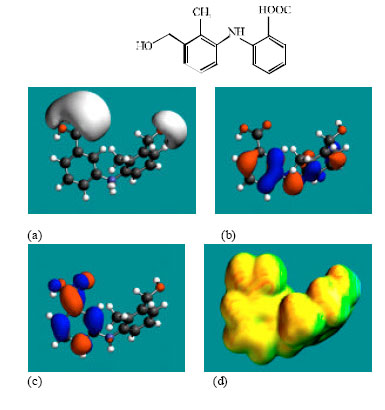 | |
| Fig. 4: | Structure of 3’HMMFA giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) surface electric charges (where red indicates negative, blue indicates positive and green indicates neutral) |
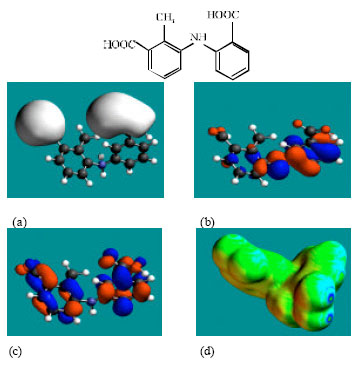 | |
| Fig. 5: | Structure of 3’CMFA giving the electrostatic potential and the HOMOs in (a) and surface electric charges in (b) where red indicates negative, blue indicate positive and green indicates neutral |
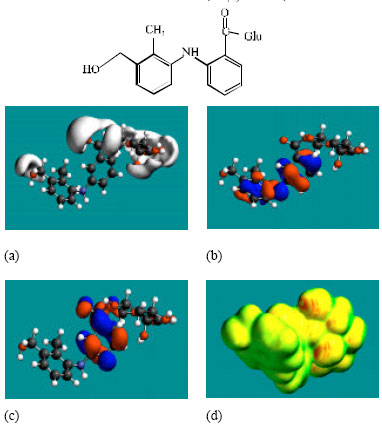 | |
| Fig. 6: | Structure of 3’HMMFA-Glu giving the electrostatic potential and the HOMOs in (a) and surface electric charges in (b) where red indicates negative, blue indicate positive and green indicates neutral |
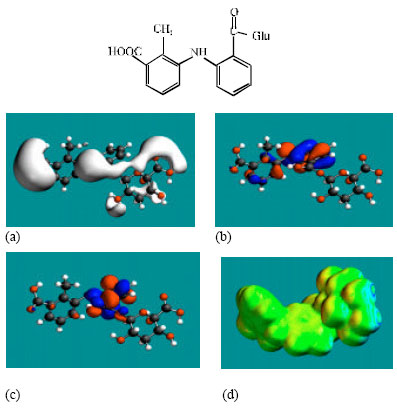 | |
| Fig. 7: | Structure of 3’CMFA-Glu giving the electrostatic potential and the HOMOs |
MFA and its metabolites have moderately low LUMO-HOMO energy differences so that they are expected to be to some extent labile. MFA-Glu has a much smaller LUMO-HOMO energy difference (3.35 eV as against 4.3 to 4.7 eV for other compounds) so that MFA-Glu would be much more labile. From quantitative structure-activity analysis it was found that for NSAIDs with a benzoic acid moiety, cytotoxicity correlated linearly with LUMO-HOMO energy gap (the smaller the gap, the greater being the toxicity) (Siraki et al., 2005). Relatively small LUMO-HOMO energy difference found for MFA and its metabolites, thus provides an explanation as to why they may be toxic. On the basis of small LUMO-HOMO energy difference, MFA-Glu would be expected to be most toxic since it would react most readily with biomolecules including intra- and extracellular proteins. However, this would contradict with the accepted idea that acyl-glucuronides are less cytotoxic. It may be that the formation of acyl glucuronide reduces toxicity associated with the carboxyl group but may introduce new ones due its higher reactivity.
In the case of MFA, 3’HMMFA, 3’CMFA and 3’CMFA-Glu the electrostatic potential is found to be more negative around the carboxyl oxygen atoms, indicating that the positions may be subject to electrophilic attack. In the case of MFA-Glu, the electrostatic potential is found to be more negative around the carbonyl oxygen atom, the amino nitrogen and the glucuronic acid moiety, indicating that the positions be subject to electrophilic attack. In the case of 3’HMMFA, the electrostatic potential is also found to be more negative around hydroxyl atom, indicating that the position may be subject to electrophilic attack. In the case of 3’HMMFA-Glu and 3’CMFA-Glu, the electrostatic potential is also found to be more negative around the hydroxyl oxygen atom, carbonyl oxygen atom and the glucuronic acid moiety, indicating that the positions be subject to electrophilic attack.
In the case of MFA, MFA-Glu, 3’HMMFA, 3’CMFA, 3’HMMFA-Glu and 3’CMFA-Glu, the HUMOs with high electron density are found close to the non-hydrogen atoms of the two phenyl rings and the linking amino nitrogen atom whereas the LUMOs are found close to oxygen atoms of the non-hydrogen atoms of the phenyl ring bonded to carboxyl group.
The abundance of neutral green and slightly negative yellow regions on the surface of MFA with some red and blue patches indicates that the interaction of MFA with biomolecules including water is more likely to be hydrophobic than hydrophilic such that MFA would have moderately low solubility in water and slightly greater solubility in lipid. It was noted earlier that MFA has a reasonably large solvation energy indicating it has moderate solubility in water. The abundance of slightly negative yellow and neutral green regions with some red patches on the surface of MFA-Glu, 3’HMMA and 3’CMA indicates that the interaction of MFA-Glu, 3’HMMA and 3’CMA with biomolecules including water would be more hydrophilic than that of MFA so that MFA-Glu, 3’HMMA and 3’CMA would be more soluble in water than MFA. The abundance of slightly negative yellow and neutral green regions on the surface of 3’HMMFA-Glu and 3’CMFA-Glu with some red indicates that the interaction of 3’HMMFA-Glu and 3’CMFA-Glu with biomolecules including water is less likely to be hydrophilic than that of 3’HMMFA and 3’CMFA.
Molecular modelling analyses show that MFA and its metabolites have moderately low LUMO-HOMO energy differences so that they would be moderately labile except MFA-Glu which has a much smaller LUMO-HOMO energy difference and hence a much greater lability. Much greater lability allows MFA-Glu to react more readily with both intra- and extracellular proteins as a result of which new types of toxicity may be introduced.
Fazlul Huq is grateful to the School of Biomedical Sciences, The University of Sydney for the time release from teaching.