Research Article
Molecular Modelling Analysis of the Metabolism of Latanoprost
School of Biomedical Sciences, Faculty of Health Sciences, The University of Sydney, Australia
Prostaglandins (PGs) contribute to numerous physiological events in mammalian tissues, including the regulation of vascular tone, reproduction, sleep, body temperature, bronchoconstriction, pain and sensitivity of photoreceptors (Samuelsson et al., 1995). Latanoprost (13,14-dihydro-17-phenyl-18,19,20-trinor-PGF2α isopropyl ester; LP) is a synthetic derivative of the natural prostaglandin PGF2α (Camras and Siebold et al., 1989; Kertstetter et al., 1988), used as an anti-glaucoma agent. It is effective in various types of glaucoma and ocular hypertension. It reduces Intraocular Pressure (IOP) mainly by increasing outflow of aqueous humor (Schachtschabel et al., 2000). LP is the active component of XalatanR eye drops, a new medication for glaucoma treatment (Sjoquist and Stjernschantz, 2002). Although systematic side effects of LP appear to be low, several ocular side effects including increase in length, number, colorization and thicknes of eyelashes and hypertrichosis have been reported in the treatment of glaucoma with LP and other prostaglandin analogues (Susanna et al., 2002; Huang et al., 2002). LP also appears to aggravate epithelial herpes and increases the risk of recurrences of herpetic keratitis (Kauffman et al., 1999). Increased iris pigmentation occurs in at least 10% of hazel-eyed patients after treatment with LP (Watson, 1999). The pathogenesis of iris darkening after topical administration of LP is not completely understood. It has been suggested that this may result from elevated melanin synthesis (Susanna et al., 2002).
LP is de-esterified by passing through the cornea to give acid form of lantaprost (LPA) that is eliminated from the eye without any further significant metabolism (Sjoquist et al., 1998). However, another potential metabolite of LP is likely to be 15-ketolatanoprost (FKLP) produced from the oxidation of the 15-hydroxyl group. FKLP which may also be de-esterified to form the corresponding keto acid (FKLPA).
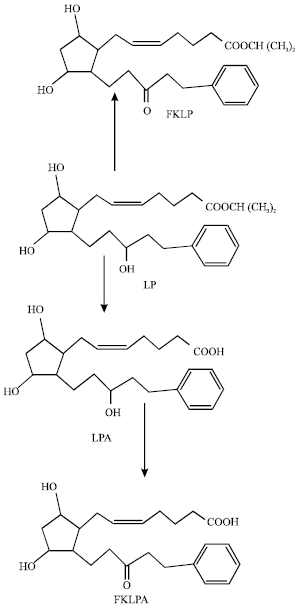 | |
| Fig. 1: | Metabolic pathways for latanoprost (Based on Kashiwagi et al., 2002) |
In this study, molecular modelling analyses have been carried out using the program (Spartan, 2002) to investigate the relative stability of LP and its metabolites with the aim of providing a better understanding on their relative toxicity. The study was carried out in the School of Biomedical Sciences, The University of Sydney during March to July 2006.
COMPUTATIONAL METHODS
The geometries of loxapine and its metabolites have been optimized based on molecular mechanics (Fig. 1) semi-empirical and DFT calculations, using the molecular modelling program Spartan ’02. Molecular mechanics calculations were carried out using MMFF force field. Semi-empirical calculations were carried out using the routine PM3. DFT calculations were carried at B3LYP/6-31G* level. In optimization calculations, a RMS gradient of 0.001 was set as the terminating condition. For the optimized structures, single point calculations were carried out to give heat of formation, enthalpy, entropy, free energy, dipole moment, solvation energy, energies for HOMO and LUMO. The order of calculations: Molecular mechanics followed by semi-empirical followed by DFT ensured that the structure was not embedded in a local minimum. To further check whether the global minimum was reached, some calculations were carried out with improvable structures. It was found that when the stated order was followed, structure corresponding to the global minimum or close to that could ultimately be reached in all cases. Although RMS gradient of 0.001 may not be sufficiently low for vibrational analysis, it is believed to be sufficient for calculations associated with electronic energy levels.
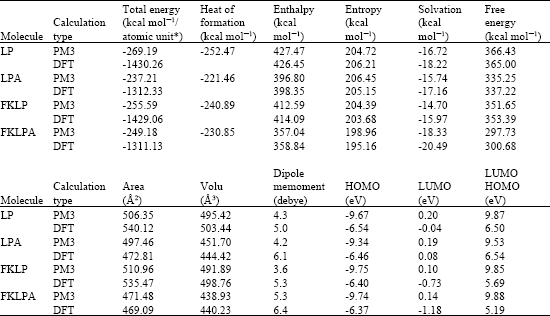
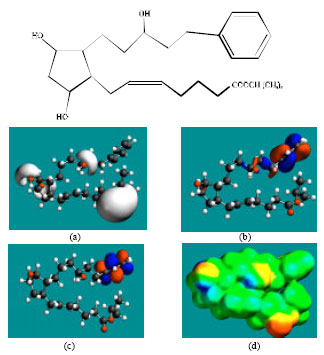
Table 1 gives the total energy, heat of formation as per PM3 calculation, enthalpy, entropy, free energy, surface area, volume, dipole moment, energies of HOMO and LUMO as per both PM3 and DFT calculations for LP and its metabolites LPA, FKLP and FKLPA. Figure 2-5 give the regions of negative electrostatic potential (greyish-white envelopes) in (a), HOMOs (where red indicates HOMOs with high electron density) in (b), LUMOs in (c) and density of surface charges (where red indicates negative, blue indicates positive and green indicates neutral) in (d) as applied to the optimized structures of LP and its metabolites LPA, FKLP and FKLPA.
The calculated solvation energies for LP and its metabolites LPA, FKLP and FKLPA from PM3 calculations in kcal mol-1 are, respectively-16.72,-15.74,-14.70 and-18.33 and their dipole moments from DFT calculations are 5.0, 6.1, 5.3 and 7.4, respectively. The values suggest LP and its metabolites would not differ greatly in their solubility in water.
LP and its metabolites are found to have large LUMO-HOMO energy differences (that range from 5.2 to 6.5 eV from DFT calculations), indicating that the compounds be kinetically inert. This means that the rate of any adverse reaction between LP and its metabolites with biomolecules such as glutathione and nucleobases in DNA is likely to be low.
In the case of LP, LPA, FKLP and FKLPA, the electrostatic potential is found to be more negative around the various oxygen centers (especially those of the ester bond in the case of LP and FKLP and of carboxyl group in the case of LPA and FKLPA), indicating that the positions may be subject to electrophilic attacks.
| Table 1: | Calculated thermodynamic and other parameters of LP and its metabolites |
 | |
 | |
| Fig. 2: | Structure of LP giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs (where red indicates HOMOs with high electron density), (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
 | |
| Fig. 3: | Structure of LPA giving in: (a) the electrostatic potential(greyish envelope denotes negative electrostatic potential), (b) the HOMOs (where red indicates HOMOs with high electron density), (c) the LUMOs (where blue indicates LUMOs) and in (d) surface electric charges (where red indicates negative, blue indicates positive and green indicates neutral) |
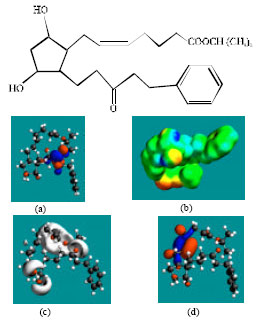 | |
| Fig. 4: | Structure of FKLP giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs (where red indicates HOMOs with high electron density), (c) the LUMOs (where blue indicates LUMOs) and in (d) surface electric charges (where red indicates negative, blue indicates positive and green indicates neutral) |
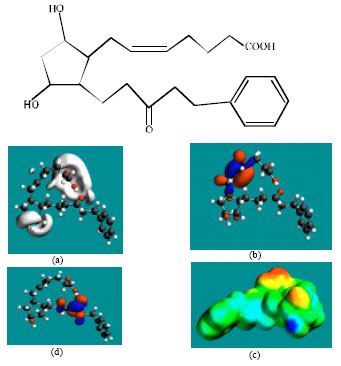 | |
| Fig. 5: | Structure of FKLPA giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs (where red indicates HOMOs with high electron density), (c) the LUMOs (where blue indicates LUMOs) and in (d) surface electric charges (where red indicates negative, blue indicates positive and green indicates neutral) |
When the surface and volume of LP are compared with those of its metabolites, it is found that the values for LP are distinctly different from those of the metabolites.
In the case of LP and LPA, both HOMOs with high electron density and LUMOs are found close to most of the non-hydrogen atoms of the phenyl ring. In the case of FKLP and FKLPA, HOMOs with high electron density are found close of some of the carbon atoms of the carbon chain containing the double bond whereas LUMOs are found close to some of the carbon atoms of the other linking carbon chain.
The overlap or close proximity of positions of HOMOs with high electron density and those of negative electrostatic potential give further support to the idea that the positions may be subject to electrophilic attacks. When the electron densities on molecular surfaces (Fig. 2d to 5d) are considered, it can be seen that the molecular surfaces of LP and all its metabolites, have both electron-rich and electron-deficient regions so that they may be subject to both electrophilic and nucleophilic attacks. The latter attack means that LP and its metabolites can react with cellular glutathione and can oxidize nucleobases in DNA. Reaction with glutathione introduces oxidative stress by compromising the anti-oxidant status of the cell whereas oxidation of nucleobses in DNA causes DNA damage. However, as stated earlier LP and its metabolites are expected to be kinetically inert so that the rates of such adverse reactions would be low.
Molecular modelling analyses based on molecular mechanics, semi-empirical (PM3) and DFT (at B3LYP/6-31G* level) calculations show that there are some electron-deficient regions on the molecular surfaces of LP and its metabolites so that they can react with cellular glutathione, thus causing glutathione depletion and hence oxidative stress and can also cause oxidation of nucleobases in DNA and hence DNA damage. However, the large LUMO-HOMO energy differences observed for LP and all its metabolites may mean that the rates of such adverse reactions may be low.
| Abbreviations | |
| Pgs: | Prostaglandins |
| LP: | Latanoprost;13,14-dihydro-17-phenyl-18,19,20-trinor-PGF2α isopropyl ester |
| FKLP: | 15-ketolatanoprost |
| DFT: | Density functional theory |
| LUMO: | Lowest unoccupied molecular orbital |
| HOMO: | Highest occupied molecular orbital |
Fazlul Huq is grateful to the School of Biomedical Sciences, The University of Sydney for the time release from teaching.