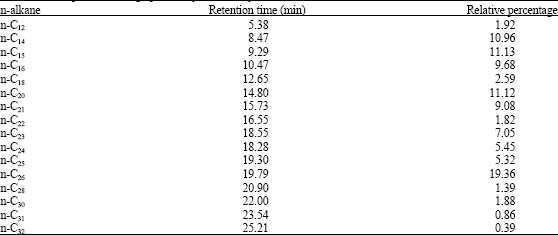
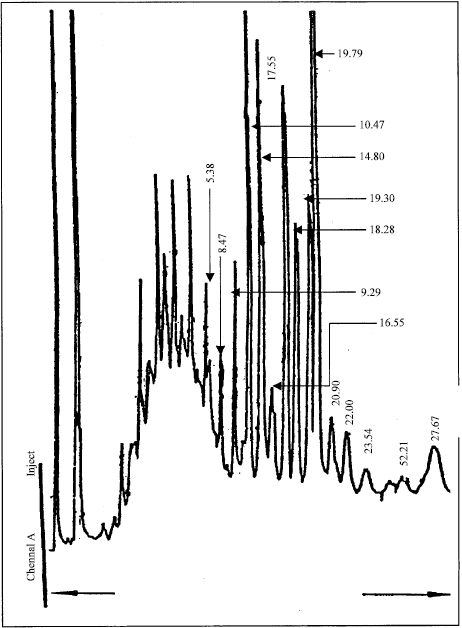
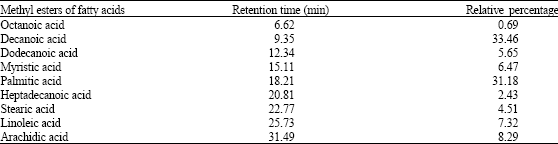
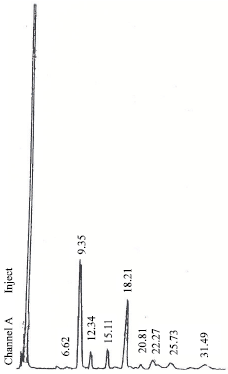
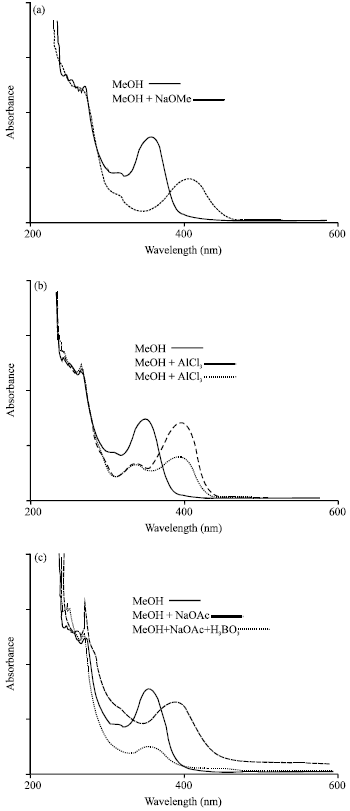
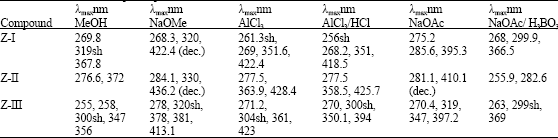
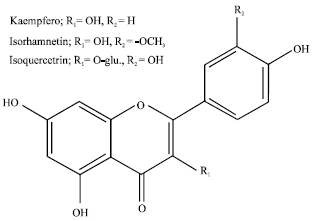
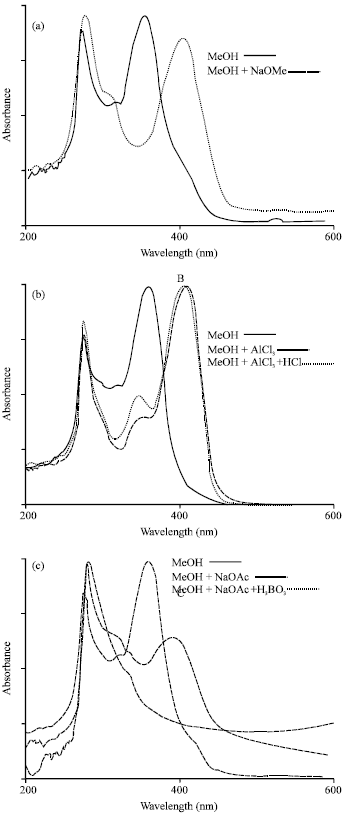
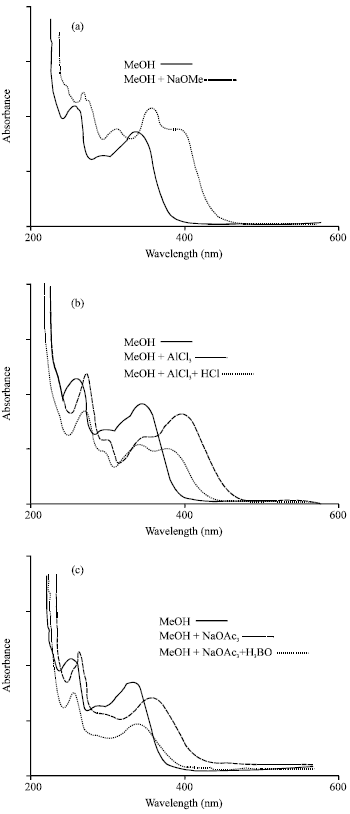
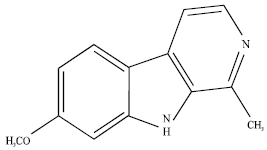
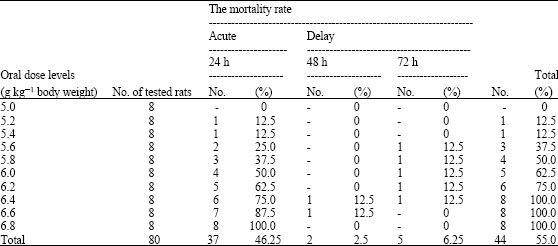
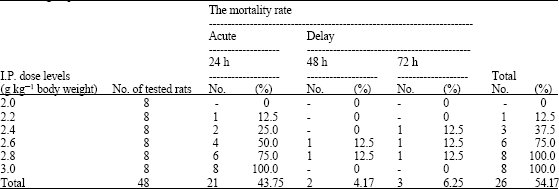
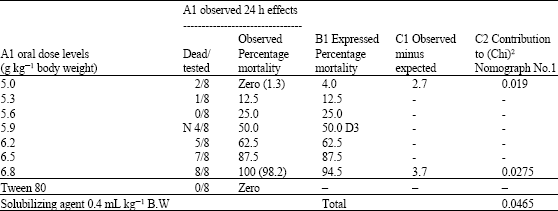
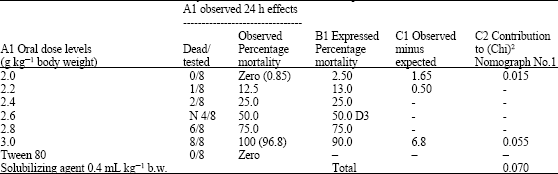
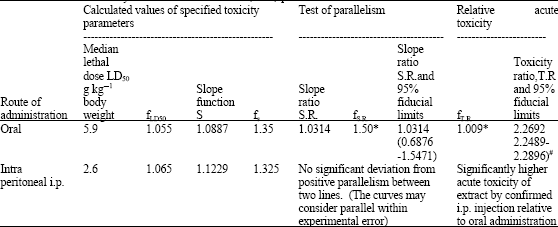
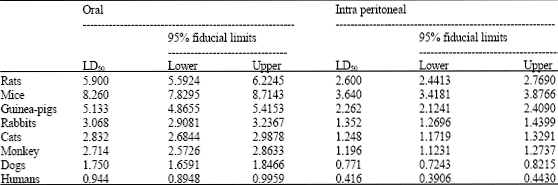
Research Article
Phytochemical and Toxicological Studies of Zygophyllum album L.f.
Department of Chemistry, Faculty of Science, Suez Canal University, Ismailia, Egypt
Ahmed I. Khodair
Department of Chemistry, Faculty of Science, Suez Canal University, Ismailia, Egypt
Faiza M. Hammouda
Chemistry of Medicinal Plants Lab, National Research Center, Dokki, Cairo, Egypt
Husseiny A. Husseiny
Chemistry of Medicinal Plants Lab, National Research Center, Dokki, Cairo, Egypt