Research Article
Molecular Modelling Analysis of the Metabolism of Chloroform
School of Biomedical Sciences, Faculty of Health Sciences, The University of Sydney, Australia
Halogenated hydrocarbons are noteworthy for their toxicity and persistence in the environment, chloroform (trichloromethane, CHCl3) is no exception. Chloroform was introduced clinically as a general anaesthetic in 1847 (Byles and Dobkin, 1971). Its use in human medicine declined because of its potential hepatic, renal and cardio-toxic properties (Pohl et al., 1980). However, chloroform is still widely used as a solvent in industrial processes and formed as a by-product during chlorination of water intended for human consumption and paper bleaching (Testai et al., 1991). Concentrations of chloroform in drinking water have been reported to range from 0.0001 to 0.3 ppm. The compound can volatilize from water and thus concentrations as high as 0.001 ppm have been reported in ambient air (Constan et al., 1999). Indoor activities such as showering or bathing as well as cooking and housekeeping may significantly contribute to total body burden of chloroform through dermal and inhalation exposure (Wallace, 1997; Backer et al., 2000). The main public health concern from chloroform exposure lies in its carcinogenic potential as it has been found to induce liver cancer in mice and renal tumours in male rats.
Epidemiological studies on populations exposed to chlorinated drinking water have shown a weak association between water consumption and cancer of bladder and lower gastrointestinal tract (Hogan et al., 1979; Cantor et al., 1998). According to Gemma et al. (2003) poor assessment of exposure and concomitant presence of many water contaminants, including other trihalomethanes and disinfection by-products, makes it difficult to establish a casual link between chloroform exposure and adverse effects in humans.
It is believed that chloroform-induced toxicity is due to its cytochrome P450-mediated bioactivation to reactive metabolites (Constan et al., 1999; Gonzalez and Butterworth, 1999). Figure 1 gives schematic representation of two metabolic pathways for chloroform in human liver-one of which follows oxidative dechlorination leading to the formation of phosgene and the other follows a reductive pathway in which a free radical is formed.
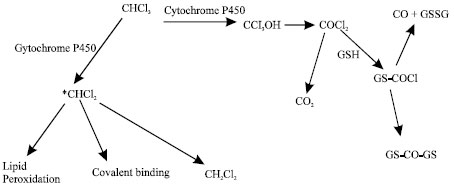 | |
| Fig. 1: | Metabolic pathways for chloroform (Gemma et al., 2003) |
The relative ratio between the two pathways depends on the relative oxygen pressure and concentration of chloroform and is found to be species-, organ- and gender-specific (Butterworth et al., 1995). At low levels, typical of actual human exposure through the use of chlorinated waters, chloroform is metabolized by high-affinity-low capacity CYP2E1 primarily to phosgene. The same enzyme is able to catalyze chloroform to dichloromethyl radical under hypoxic conditions.
Extensive in vitro and in vivo studies on rodents have shown that chloroform is oxidatively metabolized to trichloromethanol, which spontaneously decomposes to phosgene (COCl2) (Mansuy et al., 1977). The electrophilic phosgene is highly reactive and binds covalently to nucleophilic groups including proteins, phospholipid polar heads and reduced glutathione (GSH) (Pohl et al., 1981). Metabolic activation of chloroform to phosgene may be responsible for the rapid depletion of GSH observed under in vivo conditions and the subsequent liver necrosis. As glutathione plays a key role in cellular defence against oxidative stress, depletion of glutathione will induce cellular toxicity by compromise antioxidant status of the cell. Alternatively, phosgene may be hydrolysed to produce carbon dioxide and hydrochloric acid (Fig. 1).
Under anoxic or hypoxic conditions, CHCl3 may be reduced to dichloromethyl radical (*CHCl2) which is able to bind to phospholipid fatty acids or abstract a hydrogen atom from the biological environment leading to the formation of dichloromethane (Tomasi et al., 1985).
Chloroform induces tumours in rodents by a nongenotoxic-cytotoxic mode of action (Butterworth et al., 1995). Induced tumours arise secondary to events associated with cytolethality and regenerative cell proliferation. It is believed that chloroform-induced hepatotoxicity is associated with its metabolism by cytochrome P450 along the oxygen-dependent pathway resulting into its bioactivation that has been detected both in vitro and in vivo studies involving rats (Gemma et al., 2004).
In this study, molecular modelling analyses have been carried out using the programs HyperChem 7.0 (HyperChem, 2002) and Spartan ’02 (Spartan, 2002) to investigate the relative stability of chloroform and its metabolites leading to a better understanding of the toxicity of chloroform and its metabolites. Specifically the study aimed to find out whether molecular modelling analyses can provide support to the idea that one or more of the metabolites of chloroform are electrophilic in nature that are able to cause glutathione depletion. Several quantitative structure-activity relationships (QASARs models that describe a mathematical relationship between the structural features of a set of chemicals and the particular activity associated with them, have been applied to predict toxicity of chemicals including chloroform (Schultz et al., 2003; Papa et al., 2005). These are mainly based on the logarithm of octanol-water partition coefficient (logP) as a measure of hydrophobicity that determines the ability of a substance to enter cells through lipid membrane. However, such odels do not consider bioactivation e.g., the formation of electrophilic metabolites that can react with cellular antioxidant such as glutathione and can cause oxidation of nucleobases in DNA. It is believed that the molecular modelling analyses of the parent xenobiotic and its metabolites may provide more significant information about their ability to induce toxicity by causing damage to biomolecules.
Computational Methods
The geometries of chloroform, trichloromethanol, phosgene, carbon dioxide, carbon monoxide, glutathione carbonyl chloride (GSCOCl), dichloromethane radical, dichloromethane and molecular oxygen have been optimised based on molecular mechanics, semi-empirical and DFT calculations, using the molecular modelling programs Spartan ’02 and HyperChem 7.0. Molecular mechanics calculations were carried out using MM+ force field. Semi-empirical calculations were carried out using the routine PM3. DFT calculations were carried using the program Spartan ’02 at B3LYP/6-31G* level. For the optimised structures, single point calculations were carried to give heat of formation, enthalpy, entropy, free energy, dipole moment and solvation energy, HOMO and LUMO. The work was done in the School of Biomedical Sciences, The University of Sydney during October 2005 to April 2006.
Table 1 gives the total energy, heat of formation as per PM3 calculation, enthalpy, entropy, free energy, dipole moment, energies of HOMO and LUMO as per both PM3 and DFT calculations for chloroform, trichloromethanol, phosgene, carbon dioxide, carbon monoxide, glutathione carbonyl chloride (GSCOCl), dichloromethane radical, dichloromethane and oxygen. Figure 2-8 give the optimised structures of chloroform, trichloromethanol, phosgene, carbon dioxide, carbon monoxide, glutathione carbonyl chloride (GSCOCl), dichloromethane radical and dichloromethane as per PM3 calculations using the program HyperChem 7.0. The structures also give 2D contours of total electrostatic potential.
The solvation energies of chloroform, trichloromethanol, phosgene, carbon dioxide, GSCOCl, dichloromethane and oxygen from PM3 calculations in kcal mol-1 are, respectively -1.48, -5.67, -1.15, 1.87, -32.71, -1.58 and -12.58. The solvation energies of dichloromethyl radical and carbon monoxide were not provided by the program. As expected, the near terminal metabolite GSCOCl has a large value for energy of solvation indicating that the compound can be easily excreted in urine.
It was stated earlier that trichloromethanol formed along oxidative pathway is extremely reactive and spontaneously decomposes to produce electrophilic metabolite phosgene according to the equation:
| (1) |
The Gibb’s free energy (ΔG) for the reaction (1) as per DFT calculations can be shown to equal -3.65 kcal mol-1 (-5.03) - (-5.76 + 0.5 x 8.75) = -3.65), indicating that the reaction is indeed spontaneous. The relatively large LUMO-HOMO difference for CCl3OH however means that the compound would be kinetically inert if its reaction required electronic excitation. The activation energy for the reaction can however be much lower if it follows an enzymatic pathway.
As expected dichloromethyl radical has a relatively smaller LUMO-HOMO difference-smaller than that of CCl3OH-indicating that the free radical would be kinetically more labile than CCl3OH. The higher reactivity of dichloromethylradical provides support to the idea that chloroform activation occurs upon anoxic incubation with some human and ileal microsomes (Testai et al., 1991). The demonstrated in vitro metabolism of CHCl3 in human colon, but in rat colon, may indicate an association between colonic cancer and exposure to CHCl3.
| Table 1: | Calculated thermodynamic and other parameters for chloroform and its metabolites (‘DM’ stands for dipole moment) |
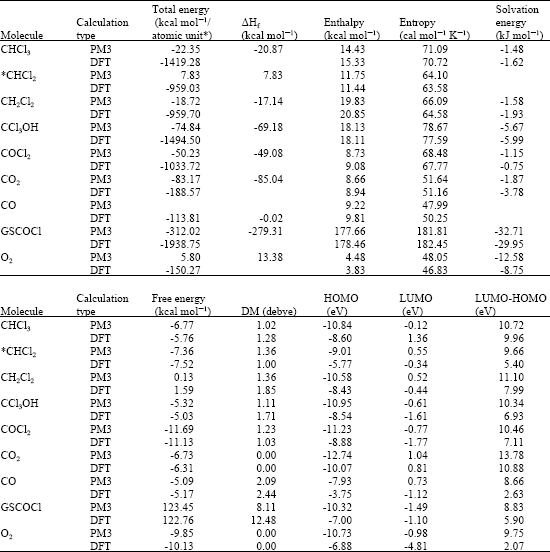 | |
| * in atomic units from DFT calculations | |
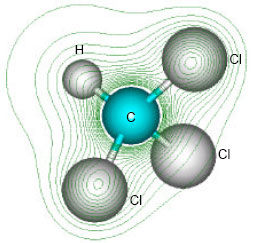 | |
| Fig. 2: | Structure of chloroform giving 2D contours of total electrostatic potential showing concentration of electron density around chlorine atoms |
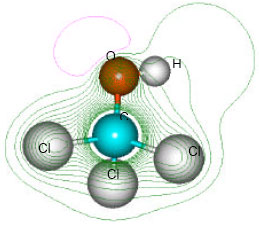 | |
| Fig. 3: | Structure of trichloromethanol giving 2D contours of total electrostatic potential showing the concentration of negative charge around the oxygen nucleus |
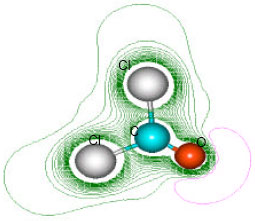 | |
| Fig. 4: | Structure of phosgene giving 2D contours of total electrostatic potential showing the concentration of negative charges around the two carbonyl oxygen atoms |
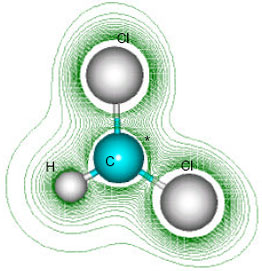 | |
| Fig. 5: | Structure of dichloromethyl radical giving 2D contours of total electrostatic potential showing the concentration of electron density around chlorine atoms |
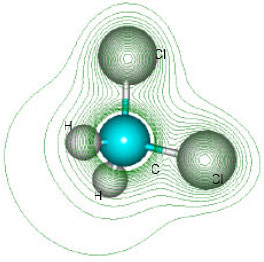 | |
| Fig. 6: | Structure of dichloromethane giving 2D contours of total electrostatic potential showing the concentration of electron density around chlorine atoms |
 | |
| Fig. 7: | Structure of carbon monoxide giving 2D contours of total electrostatic potential |
When the LUMO-HOMO energy differences of chloroform and its metabolites are compared with those of the benzene and its metabolites, it is found that the values are generally larger for the forme than the latter. This means that on relative scale chloroform and its metabolites would be less kinetically labile. The lower kinetic lability may be one reason why chloroform and its metabolites have a long persistence in the environment. This may be true also for other halogenated hydrocarbons such as DDT (Huq, 2006).
Based on LUMO-HOMO energy differences CO would be expected to be much more reactive than other metabolites of chloroform.
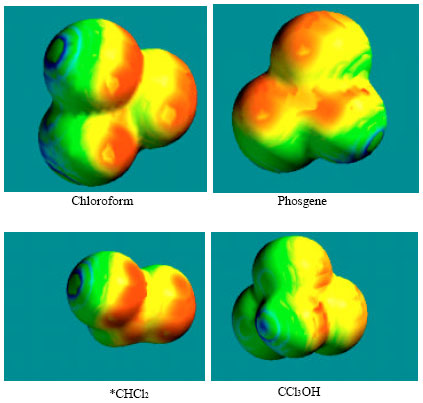
Figure 9 gives the density of electrostatic potential on the molecular surface, as applied to structures of chloroform, phosgene, *CHCl2 and CCl3OH where red indicates negative, blue indicates positive, green indicates neutral and yellow indicates slightly. It can be seen that the four compounds differ significantly in terms of distribution of surface charges. The surface of CCl3OH appears to have less negative charge although it has the highest solvation energy. Localised concentration of negative charge indicates that the four compounds may be subject to electrophilic attack.
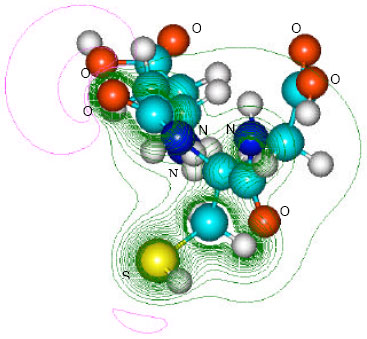 | |
| Fig. 8: | Structure of glutathione carbonyl chloride giving 2D contours of total electrostatic potential showing the concentration of negative charges around oxygens, nitrogen and sulfur |
 | |
| Fig. 9: | Structures of chloroform, phosge ne, *CHCl2 and CCl3OH giving the distribution of surface electric charges where red indicates negative, blue indicates positive, green indicates and yellow indicates slightly negative |
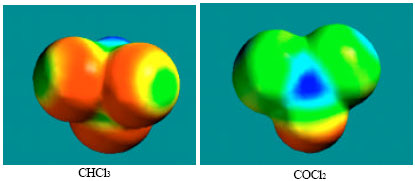 | |
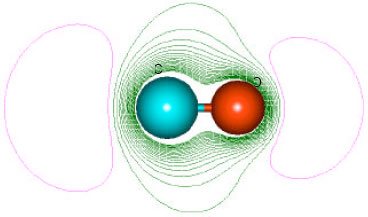
| Fig. 10: | Density of electrostatic potential on the surfaces of CHCl3 and COCl2 where blue mean electron-deficient and red indicates electron-rich and green means neutral |
However when the density of electrostatic potential is considered, it if found there are also electron-deficient regions on the surface of phosgene (Fig. 10), indicating that the metabolite can indeed react with cellular glutathione, causing its depletion and thus compromising the antioxidant status of the cell. Although the surface of CHCl3 also has an electron-deficient region, it can be seen that it abounds more in red electron-rich regions so that it may not react readily with glutathione. Also, chloroform is found to be much more inert kinetically than phosgene.
Chloroform is still widely used as a solvent in industrial processes and formed as a by-product during chlorination of water intended for human consumption and paper bleaching although it has been found to induce liver cancer in mice and renal tumours in male rats. It is believed that chloroform-induced toxicity is due to its cytochrome P450-mediated bioactivation to reactive metabolites. Molecular modelling analyses based on molecular mechanics, semi-empirical and DFT calculations suggest that the long persistence of chloroform and its metabolites in the environment may be due to kinetic inertness of the compounds. Phosgene is found to have electron-deficient regions on its molecular surface so that it can react with cellular glutathione, resulting into its depletion and thus compromising the antioxidant status of the cell.