Research Article
Molecular Modelling Analysis of the Metabolism of Modafinil
School of Biomedical Sciences, Faculty of Health Sciences, The University of Sydney, Australia
Narcolepsy is a disorder of the Central Nervous System (CNS) characterized by symptoms that include Excessive Daytime Sleepiness (EDS) with involuntary sleep episodes, cataplexy, hypnagogic hallucinations and sleep paralysis (Wong et al., 1999b). It affects about 125,000 people in the United States and appears to have a genetic basis. Modafinil (MDF, 2-[diphenylmethyl)sulfinyl] acetamide) is a unique wake-promoting agent that facilitates wakefulness in a variety of clinical models including narcolepsy, obstructive sleep apnea and shift work sleep disorder (Arnulf et al., 1997). Pateinets who are treated with antiepileptic agents frequently complain of cognitive and sedative side-effects that may limit daily function and maximum dosage in seizure management (Smith, 2003). MDF can be used to reduce such side-effects. The daily dose of MDF in the treatment of excessive daytime sleepiness in patients with narcolepsy ranges from 200 to 300 mg. Taken orally, MDF is readily absorbed reaching peak plasma concentrations within 2 to 4 h after administration. Although the exact mechanism of action of MDF is not known, it is believed to be different from those of CNS stimulants such as amphetamine or methylphenidate (Wong et al., 1999a; Engber et al., 1998).
MDF is an optically molecule with a chiral center at the sulfur atom, with both the enantiomers being equally pharmacologically active. The elimination half-life of MDF is approximately 12-15 h, which is largely reflective of the long-lived l-enantiomer. The drug is metabolized predominantly in the liver and excreted mainly as metabolites via kidney (Hellriegel et al., 2002). Metabolism is largely via amide hydrolysis and to a smaller extent by cytochrome P450-mediated oxidative pathways. The major metabolite of MDF is modafinil acid (MDFA, 2-[(diphenylmethyl) sulfinyl] acetic acid) which is not pharmacologically active. Another circulating metabolite is modafinil sulfone (MDFS, 2-[(dimethyl) sulfinyl acetamide) and a minor metabolite is phenylthio-acetic acid (PTAA). MDF is found to produce a concentration-dependent induction of CYP3A4 and CYP3A5 activity (Hellriegel et al., 2002) suggesting that there is potential for metabolic drug-drug interactions between MDF and drugs or drug metabolites that act as substrates of CYP3A4 and CYP3A5.
In this study molecular modelling analyses have been carried of MDF and its metabolites in order to obtain a better understanding of toxicity due to MDF and its metabolites. The study was carried out in the School of Biomedical Sciences, The University of Sydney during February to June 2006.
Computational Methods
The geometries of MDF and its metabolites have been optimized based on molecular mechanics, (Fig. 1) semi-empirical and DFT calculations, using the molecular modelling program Spartan ’02 (2002). Molecular mechanics calculations were carried out using MMFF force field. Semi-empirical calculations were carried out using the routine PM3. DFT calculations were carried using the program Spartan ’02 (2002) at B3LYP/6-31G* level. In optimization calculations, a RMS gradient of 0.001 was set as the terminating condition. For the optimized structures, single point calculations were carried to give heat of formation, enthalpy, entropy, free energy, dipole moment, solvation energy, energies for HOMO and LUMO. The order of calculations: molecular mechanics followed by semi-empirical followed by DFT ensured that the structure was not embedded in a local minimum. To further check whether the global minimum was reached, some calculations were carried out with improvable structures. It was found that when the stated order was followed, structure corresponding to the global minimum or close to that could ultimately be reached in all cases. Although RMS gradient of 0.001 may not be sufficiently low for vibrational analysis, it is believed to be sufficient for calculations associated with electronic energy levels.
 | |
| Fig. 1: | Metabolic pathways for MDF (Wong et al., 1999b) |
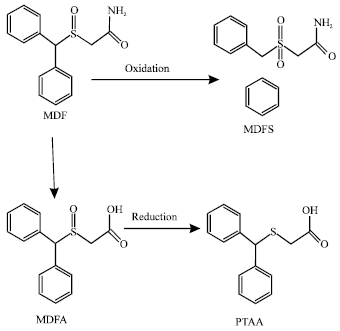
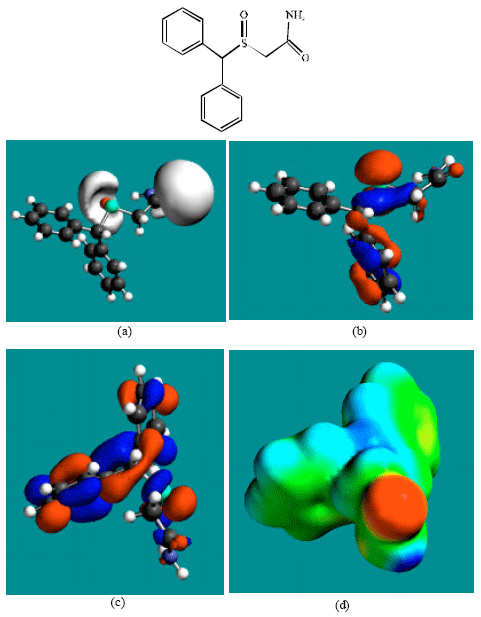
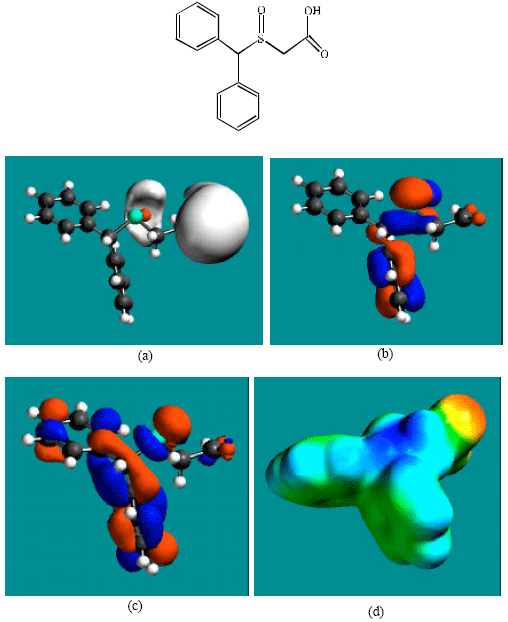
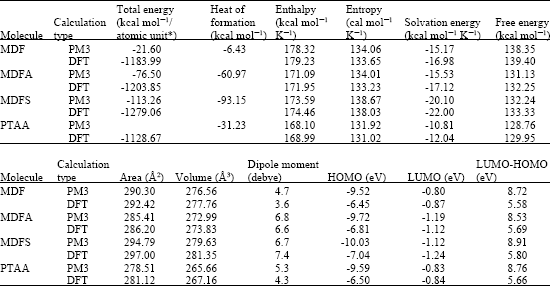
Table 1 gives the total energy, heat of formation as per PM3 calculation, enthalpy, entropy, free energy, surface area, volume, dipole moment, energies of HOMO and LUMO as per both PM3 and DFT calculations for MDF and its metabolize MDFA, MDFS and PTAA. Figure 2-5 give the regions of negative electrostatic potential (greyish-white envelopes) in (a), HOMOs (where red indicates HOMOs with high electron density) in (b), LUMOs in (c), and density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) in (d) as applied to the optimized structures of MDF and its metabolize MDFA, MDFS and PTAA.
 | |
| Fig. 2: | Structure of MDF giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
 | |
| Fig. 3: | Structure of MDFA giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
The calculated solvation energies of MDF and its metabolize MDFA, MDFS and PTAA from PM3 calculations in kcal mol-1 are, respectively -15.17, -15.33, -20.10 and -10.81 and their dipole moments from DFT calculations are 3.6, 6.6, 7.4 and 4.3, respectively. The values suggest that among MDF and its metabolites, MDFS would have the highest solubility in water and PTAA would have the lowest solubility in water.
In the case of MDF and MDFS, the electrostatic potential is found to be more negative around the sulfinyl and acetamide oxygen atoms, indicating that the positions may be subject to electrophilic attack. In the case of MDFA, the electrostatic potential is found to be more negative around the sulfinyl and carboxyl oxygen atoms, indicating that the positions may be subject to electrophilic attack.
 | |
| Fig. 4: | Structure of MDFS giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
In the case of PTAA, the electrostatic potential is found to be more negative around sulfur and acetamide oxygen atoms, indicating that the positions may be subject to electrophilic attack.
In the case of MDF and MDFA the HOMOs with high electron density are found close to most of the non-hydrogen atoms except those of one of the two phenyl rings whereas the LUMOs are found close to all the non-hydrogen atoms. In the case of MDFS and PTAA, the HOMOs with high electron density and the LUMOs are found close to most or all of the non-hydrogen atoms. The overlap of HOMO with high electron density and region of negative electrostatic potential close to sulfur, gives further support to the idea that the position may be subject to electrophilic attack.
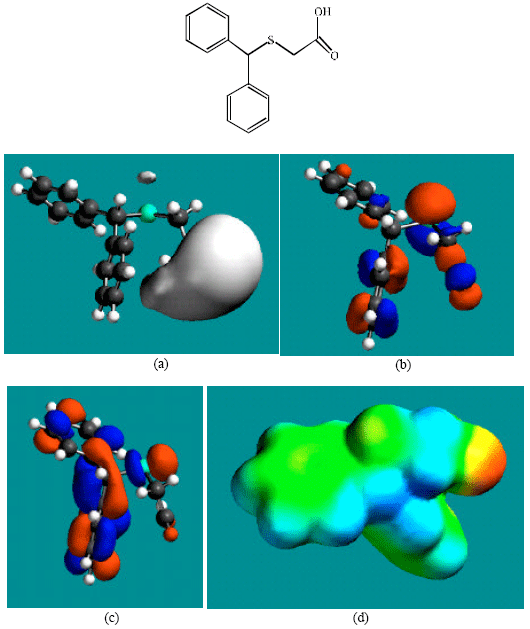 | |
| Fig. 5: | Structure of PTAA giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
When surface areas and volumes of MDF, MDFA, MDFS and PTAA are compared, it is found to that the values do not differ widely (Table 1) so that they may be able to associate with the same binding sites taken into account the dynamic nature of enzymes. In actual fact, the metabolites may not bind to the key receptors because they are not found to be pharmacologically active. This means that the difference in chemical natures of MDF and its metabolites may be the critical determinant why the metabolites may not undergo receptor binding.
| Table 1: | Calculated thermodynamic and other parameters of EP and its metabolites |
 | |
| * In atomic units from DFT calculations | |
Molecular modelling analyses based on semi-empirical and DFT calculations show that MDF and all its metabolites have large LUMO-HOMO energy differences so that they would be kinetically inert. Thus, although the molecular surfaces of MDF and its metabolites possess electron-deficient regions, the rates of reactions of the molecules with glutathione and nucleobases may be low so that MDF therapy may not be associated with cellular toxicity and DNA damage.
Fazlul Huq is grateful to the School of Biomedical Sciences, The University of Sydney for the time release from teaching.