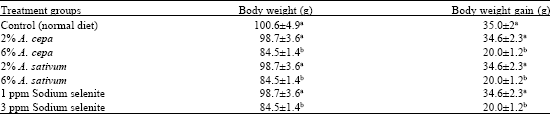
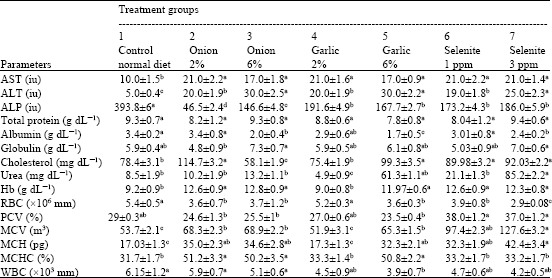
Research Article
Response of Wistar Rats to Low Levels of Dietary Allium cepa, Allium sativum and Sodium Selenite
Department of Biochemistry, School of Biotechnology, EINeelain University, P.O. Box 12702 Khartoum, Sudan
W.S. Abdel Gadir
Food Research Centre, Ministry of Science and Technology, P.O. Box 213, Khartoum North, Sudan
S.E.I. Adam
Department of Biochemistry, School of Biotechnology, EINeelain University, P.O. Box 12702 Khartoum, Sudan