ABSTRACT
Electrophysiological mechanisms of co-transmission by serotonin (5-HT) and acetylcholine (ACh) on mechanoelectrical activity of the gut were studied numerically. A mathematical model of the bursting primary sensory (AH) and motor (S) neurons linked in sequence and smooth muscle syncytium mimicked the afferent pathway of the enteric nervous plexus of the organ. The role of different receptor types, i.e., 5-HT type 3 and 4, nicotinic (nACh) and muscarinic cholinergic (μACh) and the effects of selective and non-selective receptor agonists/antagonists on the dynamics of nerve signal transduction and mechanical response in the tissue were analyzed. Results showed that selective stimulation of the 5-HT3 receptors by endogenous 5-HT reduced the threshold of activation of the mechanoreceptors by 17.6%. Conjoint excitation by serotonin of the 5-HT3 and 5-HT4 receptors located on the primary sensory (AH) and motor (S) neurons converted their regular firing pattern of electrical discharges to a beating mode. Activation confined to 5-HT3 receptors located on the somas of the AH and S type neurons, could not sustain normal electrical signal transduction between them. It required acetylcholine as a co-transmitter and a subsequent co-activation of the nACh receptors. Selective 5-HT3 receptor antagonists, e.g., Ondansetron and Granisetron, increased the threshold activation of the mechanoreceptors and inhibited dose-dependently the production of action potentials by AH neurons. 5-HT4 receptor agonists, e.g., TS-591, prucalopride and ML10302, applied alone onto the longitudinal smooth muscle of the gut did not have any effects on its electromechanical activity. However, excitation of the 5-HT4 in conjunction with μACh receptors evoked an increase in intensity of the electromechanical activity of the syncytium. GR113808A, a selective 5-HT4 antagonist, acting alone strongly inhibited smooth muscle contractions but its effect was overcome through the activation of the 5-HT3, nACh and μACh receptors. A non-selective strong 5-HT3 and weak 5-HT4 - receptor agonists, Cisapride, demonstrated a prominent effect on the AH neuron with no significant changes in the electrical activity of the S neuron. Cisapride depolarized the soma of AH neuron with the generation of high amplitude spikes. The drug caused twitch contractions of the longitudinal smooth muscle. Comparison of the theoretical results to in vivo and in vitro experimental data indicated satisfactory qualitative and quantitative agreement. The numerical investigations helped us reveal the intrinsic mechanisms of co-transmission by ACh and 5-HT and the role of receptor polymodality at the cellular and tissue levels that could not have been elucidated using the existing experimental in vivo or in vitro methods.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/jpt.2006.176.200
URL: https://scialert.net/abstract/?doi=jpt.2006.176.200
INTRODUCTION
There has been a rapid increase of experimental data on the electro-pharmacological roles of acetylcholine (ACh) and serotonin (5-HT) as neurotransmitters, the effects of their co-transmission and multiple receptor expression, in pathogenesis of various diseases. However, our understanding of the cellular mechanisms involved in the intricate pathways of processes responsible for the plethora of biological manifestations remains unsatisfactory. First, the combined processes of co-transmission cannot be studied effectively by any of the existing experimental techniques currently available to researchers for determining intrinsic mechanisms. Secondly, traditional in vivo and in vitro experimental approaches fail to provide desired quantitative information about synaptic neurotransmission. Thus, a new technique is needed that amalgamates interdisciplinary data and provides the basis for an integrated, rather than reductionist, analysis of complex biological phenomena.
Acetylcholine is a major neurotransmitter in the enteric nervous system and the gut per se. Its effects are mediated by nicotinic and muscarinic receptors. The nACh receptors are non-selective ionotropic receptors; whereas μACh receptors are part of the metabotropic - G - protein coupled receptors family. Activation of nACh receptors results in the generation of an inward calcium current with the production of fast (fEPSP) or slow (sEPSP) excitatory postsynaptic potentials, respectively. In contrast to the diverse effects of serotonin in the gastrointestinal tract, ACh always has an excitatory effect and enhances myoelectrical activity.
The ubiquitous biogenic amine 5-hydroxytryptamine (5-HT, serotonin) is present in the neurons of the enteric nervous system and to a greater extent in the enterochromaffin (EC) cells. Serotonin acts as a neurotransmitter and a paracrine messenger in the gastrointestinal tract to mediate a wide range of physiological functions. These effects are achieved through activation of 5-HT1 - 5-HT7 receptors.
A distinct neural receptor, 5-HT3, belongs to the family of ligand-gated ion channels. Serotonin applied by ionophoresis to a neuron with 5-HT3 receptors causes a short latency (< 100 ms) and a duration (< 2 s) depolarization by invoking a fast inward excitation current. The latter is due to an increase in permeability of calcium (gCa2+), potassium (gK+) and sodium (gNa+) channels.
The 5-HT4 receptors belong to a Gαs - protein - coupled family. They are positively linked to adenylyl cyclase in the second messenger signal transduction mechanism. The biological effect of their pharmacological activation is correlated with an increase in permeability of calcium-activated potassium (gCa2+-K+) and sodium (gNa+) channels and a decrease in permeability of potassium (gK+) channels.
The 5-HT3 and 5-HT4 receptors and ACh-receptors are expressed on the somas and presynaptic nerve terminals of the primary sensory (AH - type), motor (S - type) and possibly interneurons (S and AH-types) of the myenteric nervous plexus (Johnson and Heinemann, 1995). Electrochemical coupling at the neuro-neuronal synapses entails the initiation of the cascade reactions of the second messenger system and activation of ligand-operated channels. The differences in dynamics of the implicated biochemical pathways, complex allosteric neurotransmitter-receptor interactions, species and regional organ dependence, result in highly variable biological responses.
The aim of this research is to gain insight into phenomenological mechanisms of co-transmission by ACh and 5-HT and their effects on the signal transduction process in the afferent pathway of the enteric neural network of the gut. We focus primarily on the analysis of: i) the concurrent effects of co-activation of muscarinic (μACh), nicotinic (nACh) and 5-HT types 3 and 4, receptors, on the dynamics of signal transduction and ii) the role of pharmacological agents with selective, nonselective and mixed 5-HT3 and 5-HT4 - receptor agonists/antagonists mechanisms of action on the electrical activity.
The study involves mathematical concepts to model electromechanical processes of the abdominal viscera. The discussion of general physiological facts, which support the scientific concepts, mathematical formulation of the model, derivation of basic equations, construction and validation of the numerical algorithm, software design, have been described by Miftakhov and Christensen (2001), Miftakhov et al. (1999 a,b).
MATERIALS AND METHODS
A model of the bursting AH and S type neurons linked in sequence is based on the Hodgkin-Huxley formalism and includes activity of the voltage-dependent Na+, K+, Ca2+, Ca2+-K+ and leak Cl¯ channels (Tuladhar et al., 1997; Bertrand et al., 2000; Van Hooft et al., 1998). The dynamics of the propagation of electrical signals along the unmyelinated axons satisfies the classical Hodgkin-Huxley model. The synapse and the drug - receptor interaction models are equivalent to the pharmacodynamic model proposed by Miftakhov and Christensen (2001).
As a tool in our theoretical investigations we exploit a novel computational platform, Gut Discovery© (www.aincompany.com). A one-dimensional model that is composed of the interconnected primary sensory (AH) and motor (S) neurons and the smooth muscle syncytium is designed to analyze the electromechanical events in the gut. The model reproduces the following sequence of electrophysiological processes in the afferent pathway and smooth muscle syncytium: i) deformation of the free nerve endings of the mechanore-ceptors by mechanical stimuli of a known intensity and duration; ii) generation of action potentials (AP) and their propagation along the unmyelinated fiber towards the soma of the AH - neuron; iii) the action potential generation at the soma of the AH - neuron and the propagation of the wave of depolarization along the nerve axon towards the soma of the S - neuron; iv) electrochemical coupling at the axo-dendritic synapse on the soma of the secondary neuron and generation of the fEPSP; v) discharge of the soma of the S - neuron and the propagation of the wave of depolarization along the nerve axon towards the neuro-muscular synapse; vi) electrochemical coupling at the synapse and fEPSP generation; vii) activation of L-type Ca2+ channels of the smooth muscle membrane; viii) active force generation and deformation of smooth muscle. The standard parameter settings depend on the identity of the cells types and can be obtained from Miftakhov et al. (1999 a,b).
With the model we study the effects of 5-HT and ACh alone and as a result of their interactions through the process of co-transmission and co-activation of different receptor types. The effect of drugs is achieved by varying conductances for affected channels or parameters involved in the second messenger system pathway. Electrophysiological and mechanical parameters of the model are adjusted during each experiment to reproduce the species variability.
RESULTS
Electrical Activity of the Mechanoreceptors
Physiological Conditions
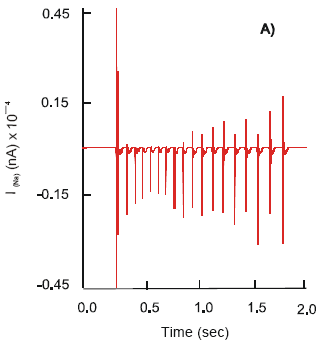
Under normal physiological conditions, deformation (ε) of the wall of the gut with multiple stretches, ε = 0.17, n = 10, of duration, td = 0.9 s, followed at intervals, Δt = 1.5 s, initiates the voltage-dependent inward sodium, INa, outward potassium, IK, currents and leak chloride, ICl, current at the free nerve endings of the mechanoreceptors. The sodium influx has an average rate 1 nA s-1 and reaches the maximum amplitude 1.14 nA (Fig. 1a). It has almost instant recovery phase, during which the strength of the current reduces to 0.23 nA, followed by a prolonged period, 0.8 s, of slow decline to the resting value: min INa = 0.042 nA. An outward K+ current shows longer duration. The dynamics of activation, IK, is linear with the exponential inactivation (Fig. 1b). The current rises to its maximum, IK = 0.04 nA, at a constant rate 0.04 nA s-1 and declines rapidly to a level 0.075 nA. The balanced activity of the ion currents, results in the production of the dendritic action potentials (APs) (φd0) at the free nerve endings of the mechanoreceptors. They have maximum amplitude 15.7 mV and duration ~ 2.3 ms.
Effect of 5-HT3 Receptors
The mechanical deformation causes the release of endogenous 5-HT from the EC cells. Free serotonin binds to the 5-HT3 receptors on the free nerve endings of the mechanoreceptors with a subsequent increase in permeability of ligand-gated ion channels. The INa current demonstrates nonlinear exponential behavior with an initial rapid rise, at a rate 2.04 nA s-1, followed by a slow dynamic, 0.2 nA s-1 (Fig. 1b). It reaches max INa = 1.77 nA. The recovery phase of the channel remains unchanged from that seen above. The dynamics of potassium channel activity have an exponential phase of activation with a slow start at the beginning, 0.05 nA s-1 and a quick climb, 0.3 nA s-1, to the maximum value, 0.188 nA (Fig. 1a). In the presence of endogenous serotonin the process of inactivation of the IK goes on faster, 0.6 nA s-1. The above change in the ionic currents reflects an increase in the amplitude of APs to 26 mV.
Presence of endogenous 5-HT reduces the threshold for mechanical activation of the mechanoreceptors by 17.6%. Multiple stretch impulses of ε = 0.14 applied to the wall of the gut are sufficient to initiate the cascade of electrical events as described above.
Effect of Selective 5-HT3 Receptor Antagonists
Treatment of the mucosa of the gut with selective 5-HT3 receptor antagonists, Ondansetron (GlaxoSmithKline) or Granisetron (Roche), abolishes the potassium current (IK ~ 0 nA). There is a significant reduction in the sodium influx. The INa current shows an activation rate of 0.16 nA s-1 and reaches the maximum amplitude of 0.38 nA. As a result, the dendritic action potentials of the amplitude 3 mV are generated. They are not strong enough to set off electrical signals at the free nerve endings and to sustain their propagation of along the unmyelinated fibers. However, an increase in the intensity of the mechanical stimulus ε by 35% (ε = 0.23) results in an unexpected response from the mechanoreceptors. There is an increase in both the INa and IK currents: max INa = 1.04 nA and max IK = 0.2 nA, are recorded (Fig. 1a,b). The mechanoreceptors produce action potentials: φd0 = 22.9 mV.
Electrical Activity of the Primary (AH) Neuron
Stimulation of Mechanoreceptors
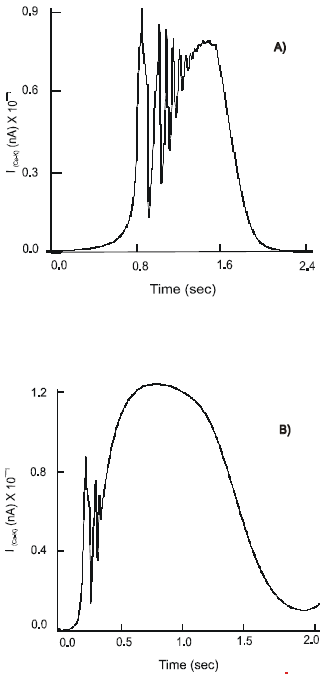
The dendritic action potentials elicited at the free nerve endings propagate along the unmyelinated axon and reach the soma of the neuron. There they activate the voltage-dependent Na+, K+, Ca2+, Ca2+-K+ and leak Cl¯ channels. The dynamics of the inward IpNa and outward IpK currents demonstrate high frequency oscillatory behavior with fast alternations of activation and inactivation (Fig. 2a). The maximal currents, IpNa = 81.5 nA and IpK = 29.8 nA are registered immediately after excitation of the membrane. In 0.2 s both currents reduce in strength: IpNa = 40.8 nA and IpK = 15 nA are recorded. The sodium and potassium channels remain active for 0.8 s.
The dynamics of activation of Ca2+ channels has slow initial phase, 1.9 nA s-1 (Fig. 2b). It is followed by a fast phase, 14.8 nA s-1, when the maximum value, 11.65 nA, is reached. There are high frequency oscillations, 0.8-3.8 (nA), in the current behavior that occur when the channel starts approaching its peak of activation.
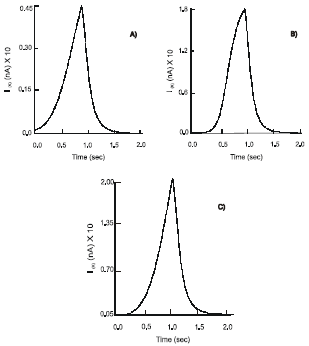 | |
Fig. 1a | |
 | |
Fig. 1b | |
| Fig. 1 a,b: | Dynamics of the IK and INa ion currents at the free nerve endings - mechanoreceptors, under normal conditions (A); after activation of the 5-HT3 receptors (B) and; following application of the selective 5-HT3 receptor antagonists (C), respectively |
The recovery of the IpCa shows the linear dynamics at a rate ~ 23 nA s-1. A similar pattern is seen with the outward IpCa-K. It quickly attains the maximum level and sustains it for 0.6 s. The max IpCa-K = 10 nA. There are high fluctuations of the current, which are only present during activation of the channel.
In absence of the free endogenous serotonin the AH neuron responds only to odd excitatory signals arriving from the mechanoreceptors with the bursts of action potentials, φ1, of the maximum amplitude 90 mV (Fig. 2c). The pattern of discharges resemble a bursting chaos type of electrical activity at a variable frequency v=8-10 (Hz).
Effect of 5-HT3 Receptors
A selective stimulation of the 5-HT3 receptors by 5-HT has a profound effect on activity of all ion channels. The duration of the active state of the Na+ and K+ channels increases to 1.54 s. There is a significant rise in intensity of the currents: max IpNa = 154.3 nA and max IpK = 52.3 nA. The brisk increase in the currents is followed by a lengthy period of recovery when fluctuations of the currents of average amplitudes, 61.7 nA and 35 nA, respectively, are observed.
The amplitude of calcium activated potassium current is: max IpCa-K = 14 nA. It demonstrates a slight increased activation rate, 25 nA s-1. The inactivation phase of the IpCa-K remains intact from the one considered above (Fig. 4a). There are no changes in the IpCa. However, the channel sustains its active state for a longer period.
As a result of the above alterations the primary sensory neuron generates max φ1= 111.3 mV. It is important to note that the neuron requires a lower level of deformation, min ε = 0.14, to responds with the production of action potentials to each impulse of excitation arriving from the free nerve endings. Its electrical pattern of discharges resembles beating mode with a frequency 7 Hz.
Effect of Selective 5-HT3 Receptor Antagonists
Ondansetron and Granisetron applied separately completely abolish any electrical activity of the neuron. The neuron remains silent after external periodic stimulations of intensities, 0.14 - 0.17, at the free nerve endings of the mechanoreceptors. However, larger deformation, ε = 0.24, causes an unexpected production of action potentials of short duration, 0.3 s and max φ1 = 91 mV. A gradual increase in the concentration of the selective 5-HT3 antagonists has a dose-dependent effect on the reduction of excitability of the neuron.
Effect of 5-HT4 Receptors
Excitation of the 5-HT4 receptors on the soma of the primary sensory neuron by the free serotonin results in the production of high amplitude, IpNa = 166.6 nA and IpK = 46.1 nA and short duration, 0.2 s, sodium and potassium currents. The Na+ channel returns quickly to its unexcited state, while K+ channels require ~ 1 s to recover. The calcium current shows fast activation, at a rate of 36 nA s-1 and reaches the maximal amplitude, max IpCa =21.7 nA. The dynamics of its recovery has two distinct phases: the initial phase with a rate 5.6 nA s-1 and the faster phase - 8.64 nA s-1. The IpCa-K shows a relatively smooth rise to the maximum value, 12.6 nA. The IpCa-K remains active for ~ 2.55 s (Fig. 2a).
There is an increase in depolarization of the soma with a shift of the resting membrane potential to 50 mV. The neuron fires a few high amplitude APs, max φ1 = 96.2 mV, followed by a slow process of decay of depolarization. There is also a reduction in duration, 0.6 s, of the period of after-hyperpolarization.
Effect of Co-expression by 5-HT3 and 5-HT4 Receptors
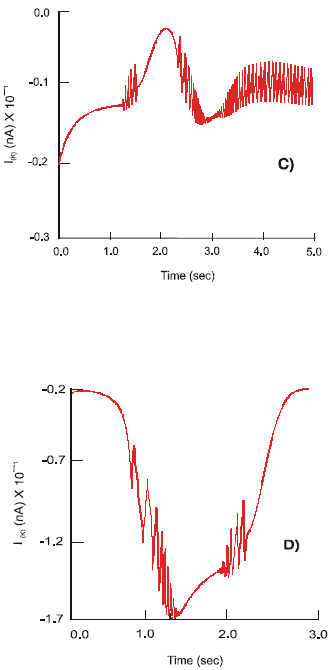
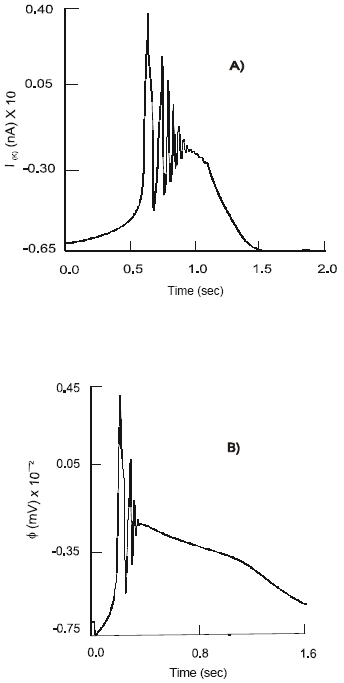
A concomitant stimulation of the co-localized 5-HT3 and 5-HT4 receptors on the soma of the primary neuron results in increase in its excitability. At the beginning of the process, when activity of the ligand-gated 5-HT3 receptors dominates, the currents of amplitude IpNa =25 nA and IpK = 36 nA are recorded. As a result of a full affect of the 5-HT4 receptors the dynamics of the sodium and potassium channels change to a high frequency oscillatory mode. Ion current of intensity IpNa = 45 nA and IpK = 12 nA are generated. The Ca2+ channel reiterates the type of activity observed under a separate stimulation of the 5-HT3 receptors. With concurrent excitation of the 5-HT4 receptors the pattern of the IpCa changes to a beating mode with constant amplitude of fluctuations, 4 nA (Fig. 2b).
The neuron produces spikes of high amplitude, max φ1 = 81 mV, followed by a long quiescent period of sustained depolarization: -13 < φ1 < 25 mV. This response is characteristic of a separate activity of the fast ligand-gated 5-HT3 receptors. A transitory period of irregular chaotic type discharges of APs corresponds to the gradual activation of the 5-HT4 receptors. Amplitude of φ1 increases from 5 to 85 (mV) and the frequency of firing varies between 18 < v< 37 (Hz). As the 5-HT4 receptors become fully active the pattern of electrical activity of the soma begins to resemble a beating mode. Action potentials of average amplitude φ1= 50 mV and a frequency v = 28 Hz are generated.
The selective 5-HT3 receptor antagonist, Ondansetron, abolishes the production of spikes on the soma. Continuous activation of the 5-HT4 receptors results in a translation of the electrical pattern of the neuron from the beating to a self-excitatory mode. Regular rhythmic undulations of the membrane potential of amplitude 30 mV with single spikes, max φ1 = 40 mV, on crests of the waves are produced. This effect persists while the 5-HT4 receptors remain active.
Effect of Cisapride
A non-selective strong 5-HT3 and weak 5-HT4 - receptor agonists, Cisapride, increases the influx of calcium ions into the cell. The maximal amplitude of the IpCa equals 17.2 nA. There are changes in the patterns of activity of the IpNa, IpCa-K and IpK currents with a period of irregular high frequency oscillatory activity and an interim quiescent period. Thus the maximal values of the IpNa and IpK of 228.5 nA and 57.6 nA, respectively, are achieved immediately after the application of Cisapride. The second peak of activity of the duration 0.5 s shows max IpNa = 38 nA and IpK = 18.8 nA. The IpCa-K current demonstrates a steady rise in amplitude with max IpCa-K = 13 nA at the beginning of the process followed by the second peak of IpCa-K = 19.2 nA (Fig. 2a).
Application of Cisapride depolarizes the soma of the primary neuron and elevates the resting membrane potential: φ1(rest) = - 62.5 mV. The neuron generates spikes of high amplitude, max φ1 = 111 mV and frequency, 7 ÷ 18 (Hz). There is a period of 0.65 s when no action potentials are produced. It is followed by a short period of bursting activity with max φ1 = 62 mV and duration 0.5 s.
Electrical Activity of the Motor (S) Neuron
Effect of nACh Receptors
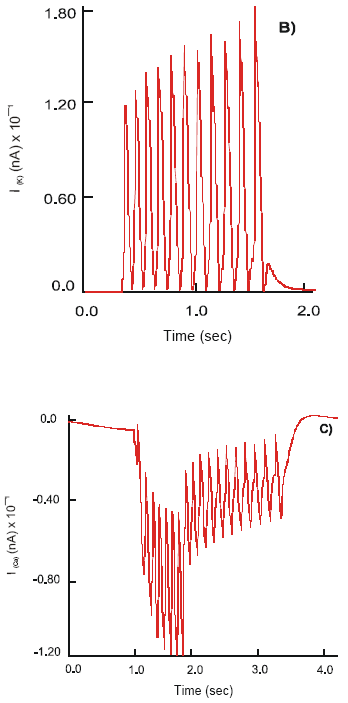
Depolarization of the presynaptic membrane at the neuro-neuronal synapse activates release of vesicular acetylcholine and its diffusion into the synaptic cleft. The main part of ACh reaches the postsynaptic membrane and reacts with the receptors on the soma of the motor neuron. The generation of fEPSP starts with the beginning of (ACh-R)-complex development. It increases as a step function and achieves its maximum 87.1 mV in 0.25 ms. The level of fEPSP is sufficient to excite the neuron. It discharges a single train of pulses of amplitude 102.1 mV at a frequency8.4 Hz and duration 1.9 s. The observed pattern of electrical activity resembles a regular bursting mode.
 | |
 | |
 | |
 | |
 | |
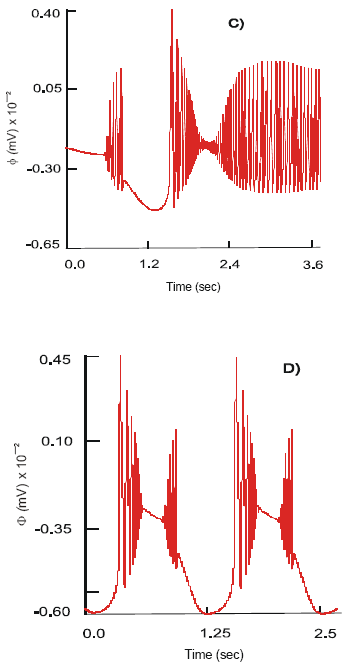 | |
| Fig. 2 a,b,c: | Dynamics of the IpCa – K , IpCa ion currents and action potentials, φ , on the soma of the primary (AH) sensory neuron after selective stimulation of: 5-HT3 receptors (A); 5-HT4 receptors (B); concomitant stimulation of the 5-HT3 and 5-HT4 receptors (C) and after treatment with Cisapride (D) |
 | |
 | |
 | |
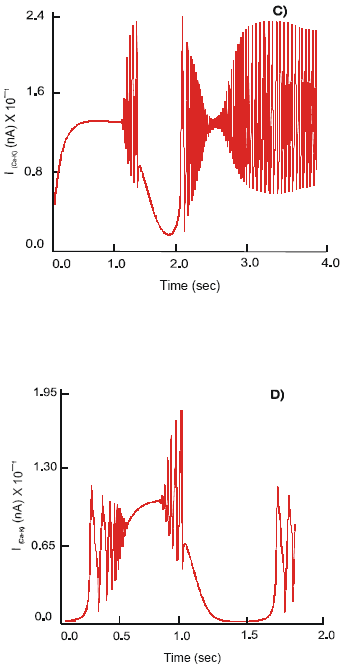
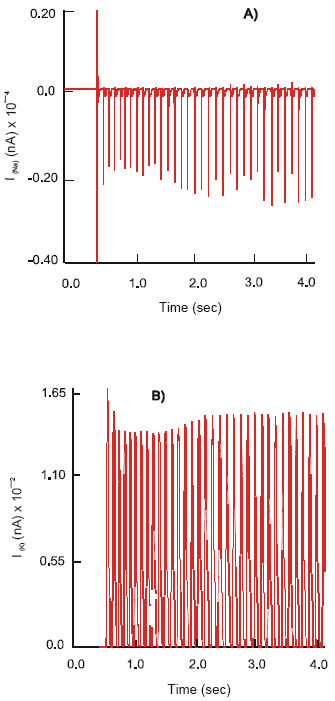
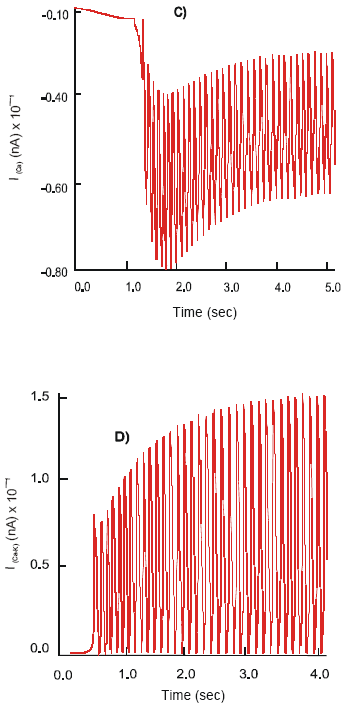
| Fig. 3: | Dynamics of the ImNa (A), ImK (B), ImCa (C) and ImCa (D) ion currents and action potential, (E), φ on the soma of the motor (S) neuron after selective stimulation of the 5-HT4 receptors |
 | |
 | |
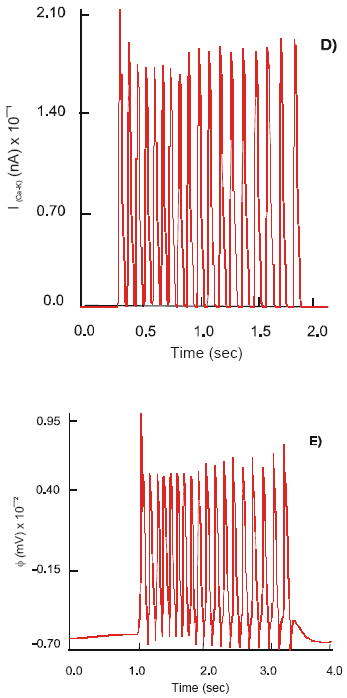 | |
| Fig. 4: | Dynamics of the ImNa (A), ImK (B), ImCa (C) and ImCa (D) ion currents and action potential, (E), φ on the soma of the motor (S) neuron after co-joint stimulation by 5-HT and ACh of 5-HT3 and nACh receptors |
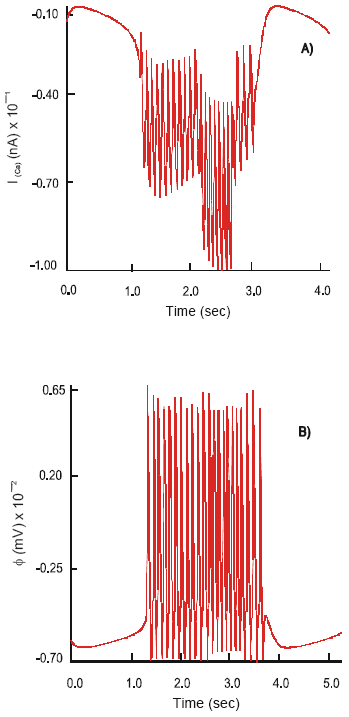 | |
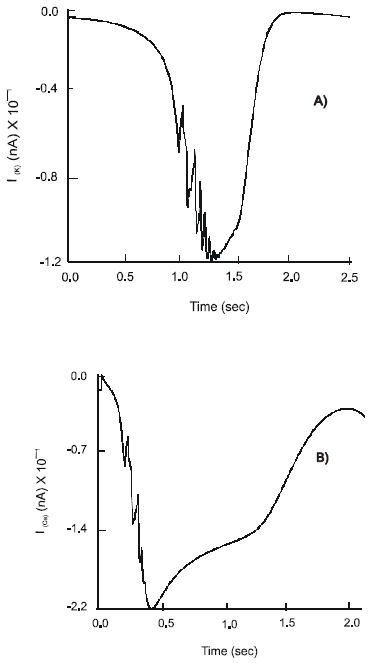
| Fig. 5: | Changes in the dynamics of the ImCa(A) ion current and action potential,φ(B), on the soma of the S-neuron after application of Cisapride and co-stimulation of the nACh receptors |
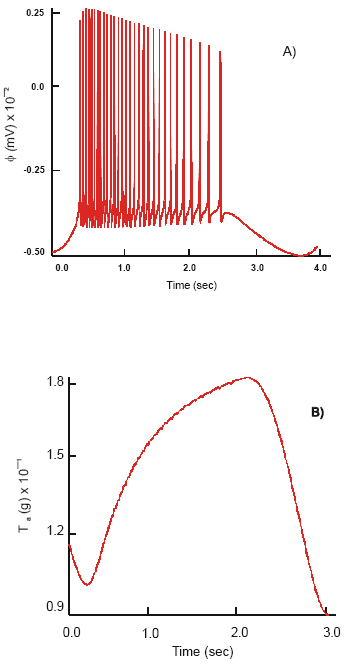 | |
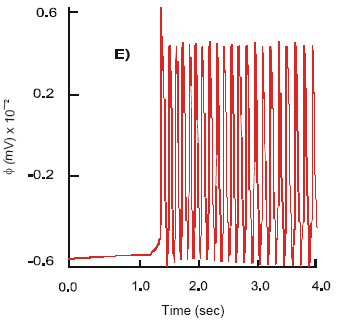
| Fig. 6: | Changes in slow wave, φ (A) and active force, Tal (B), dynamics in the longitudinal smooth muscle after co-joint activation of the 5-HT4 and μACh receptors |
Effect of the 5-HT3 and 5-HT4 Receptors
A selective stimulation of 5-HT3 receptors by the free endogenous 5-HT in absence of ACh in the system does not have excitatory effect on the soma of the motor neuron.
In contrast, activation of the 5-HT4 receptors changes the pattern of neuronal electrical activity into a long lasting excitatory mode. Action potentials, φ2, of constant amplitude 114 mV and a frequency 7 Hz are produced. The ion currents: ImNa of magnitude 22,5x102 nA, ImK ~ 148 nA, ImCa-K ~ 16.2 nA and ImCa ~ 6 nA are recorded (Fig. 3).
Effect of Co-activation of the 5-HT3 and nACh Receptors
Simultaneous excitation of the 5-HT3 and nΑCh receptors on the soma of the motor neuron results in its hyperpolarization, φ2(rest) = 66.8 mV. The neuron generates action potentials of the maximal amplitude 134 mV at a frequency 7.8 Hz. The observed electrical activity is due to an increase in the dynamics of ion currents: ImNa = 18.5x102 nA, ImK = 18 nA and ImCa-K = 14.8 nA. They demonstrate a regular high frequency mode of activation - inactivation. The calcium current shows an intensive oscillatory phase of activation followed by a slow recovery period. Max ImCa = 11.9 nA is recorded (Fig. 4).
Effect of co-activation of the 5-HT4 and nACh Receptors
Concurrent stimulation of co-localized 5-HT4 and nACh receptors initiates strong large magnitude sodium ion current, max ImNa = 27x102 nA. There is a concomitant slight decrease in amplitude of the ImK, ImCa-K and ImCa currents, compare to a separate excitation of 5-HT4 and/or nACh receptors. These changes lead to the generation of the spikes of amplitude, φ2=116.5 mV.
Effect of Selective 5-HT3 and 5-HT4 Receptor Agonists and the Co-expression of nACh Receptors. Cisapride and renzapride added into the system have a pronounced effect on the dynamics of the calcium current. Thus max ImCa = 7.04 nA is observed. The Ca2+ channel shows oscillatory behavior with fluctuations of the current of amplitude 4.2 nA and constant frequency: v = 6.4 Hz. Ion currents of intensity: ImK = 210 nA, ImCa-K = 16.7 nA and ImNa = 28.5x102 nA are recorded. The soma of the neuron is hyperpolarized to -70 mV. The neuron remains in a highly excitable state for the duration of 3.44 s and generates APs max φ2 = 130 mV and a frequency of v = 6.4 Hz.
No significant changes are observed in the pattern of behavior of ImK, ImCa-K and ImNa ion currents after simultaneous application of Cisapride and the release of ACh into the system. However, the dynamics of the ImCa demonstrates two distinct peaks of activation (Fig. 5). Immediately after activation of the 5-HT3, 5-HT4 and nACh receptors max ImCa = 6.8 nA is observed. The current slightly decreases in intensity to 6.45 nA during the following 1.2 s. The second maximum of the influx of calcium ions with max ImCa = 9.5 nA is recorded 1 s later. The current quickly subsides to its inactive state.
Interestingly the duration of electrical excitability of the neuron reduces to 2.8 s. There is a decrease in the maximal amplitude of APs, max φ2 = 120 mV and an increase in frequency of spikes: v = 7.8Ηz. The level of hyperpolarization of the soma remains unchanged.
Effects of Selective 5-HT3 - Receptor Antagonist and Co-expression of nACh Receptors
Ondansetron and Granisetron block the serotonergic pathway between the primary sensory and motor neurons. The normal electrical signal transduction is maintained through the co-existing cholinergic mechanisms. The latter sustain the neuro-neuronal synaptic connectivity. They are responsible for the generation of short duration (< 100 ms) and 85-90 mV in amplitude fEPSP on the soma of the adjacent motor neuron.
Electromechanical Activity of Smooth Muscle
Effect of μACh receptors. fEPSP at the neuro-muscular synapse activate the L-type Ca2+ channels located on the smooth muscle membrane. Alterations of the permeability of the channels cause cyclic transitory changes in the myoelectrical pattern. The slow wave mode transforms to bursting chaos with the generation of fast APs at a frequency of v = 17 Hz. Their amplitude is 23 mV at the beginning and decreases towards the end of the burst, φm = 8 mV. The bursting chaos transforms to regular bursting with generation of spikes on the crests of slow waves. The burst amplitude, burst duration and number of spikes per burst all increase. The action potentials have maximum amplitude of 72 mV and oscillate at a maximum frequency of 19 Hz. The regular bursting converts back to bursting chaos and slow wave mode.
There is a gradual increase in the concentration of intracellular calcium ions, mainly due to the influx of ions through the activated slow Ca2+ channels. Maximum internal calcium concentration ([Ca2+]i = 0.46 μM) is achieved. As a result of activation of the contractile protein system, rhythmic contractions of the longitudinal smooth muscle, max Ta = 14.4 mN cm-1, are produced. The duration of electromechanical activity is 2.6 s.
Effect of 5-HT4 Receptors
Selective 5-HT4 receptor agonists, TS-591, prucalopride and ML10302, applied onto the longitudinal smooth muscle of the gut do not have any effects on its electromechanical activity.
Effect of co-activation of the 5-HT4 and μACh Receptors
Excitation of the 5-HT4 receptors with TS-591, prucalopride and ML10302 in conjunction with μACh receptors evokes an increase in intensity of the membrane ionic currents and electromechanical activity of the smooth muscle syncytium. The ILCa demonstrates high frequency alternations, 18.5 Hz, of short duration. Average amplitude of the L-type Ca2+ current is 7 nA and max ILCa = 10.6 nA. The dynamics of the transitory calcium current, ITCa, remains unchanged. It oscillates at a frequency 21 Hz and has average amplitude 0.30 nA. The outward voltage-dependent K+ and Ca2+ - K+ currents are reduced in strength. Maximum IsCa-K = 1.37 nA and IsK = 4.6 nA are recorded. The above changes in membrane ionic currents cause an increase in the frequency of the production of APs, v = 20 Hz. The amplitude of spikes is slightly decreased, φm = 68 mV, compare to that observed when only μACh receptors are active.
The concentration of free cytosolic calcium is 0.46 μM. As a result, the longitudinal smooth muscle responds with the production of phasic contractions. The maximum active force (T a) generated equals 15.6 g. The contractions are of duration 3.45 s, concurrent in phase and time with the dynamics of the intracellular Ca2+ oscillations and have constant amplitude, 6 g. The calculated velocity of twitch is 5.8 g cm-1 (Fig. 6A and B).
Effect of Selective 5-HT4 Receptor Antagonists and the Co-activation of the 5-HT3 and μACh Receptors Addition of the selective 5-HT4 antagonist, GR113808A, strongly inhibits contractions evoked by the free endogenous serotonin. However, electromechanical activity can be sustained through the function of the neuronal 5-HT3 and nACh receptors and μACh receptors located to the smooth muscle membrane. Thus, application of Cisapride leads to twitch contractions of smooth muscle of amplitude Ta = 13.8 mN cm-1.
DISCUSSION
With our experiments we were able to reproduce numerically the pharmacological patterns of behavior and to study the interplay between ACh and 5-HT. We analyzed systematically their exact effects on electrical activity of the primary sensory (AH) neuron and the motor (S) neuron in the afferent neural pathway of the enteric nervous plexus.
Comparison of our numerical results to experimental data shows satisfactory qualitative and quantitative agreement. Thus, using the isolated guinea pig ileum, Tuladhar et al. (1997; 2000) convincingly demonstrated that activation of the 5-HT3 receptors by mucosally applied free 5-HT reduces the mechanical threshold for initiation of the peristaltic reflex by 20-26%. Present results show a 17.6% decrease from the required min ε. Further authors showed that treatment of the preparation with selective 5-HT3 receptor antagonists, Ondansetron and Granisetron, resulted on an average 44% increase in the level of mechanical stimulus needed, which is in a satisfactory agreement with our findings - 35%. Present results indicate that a possible mechanism is associated with an increase in the conductivity of the Na+ and K+ channels and changes in the dynamics of activation of the INa and inactivation of the IK currents. Interestingly, the inhibitory effect of Ondansetron and Granisetron with a complete suppression of the potassium current (IK ~ 0 nA) and a significant reduction in strength of the INa current can be overcome by a higher level of mechanical deformation. This fact suggests that the 5-HT3 receptors may be directly linked to the mechanosensitive Na+ channels or are located in close proximity to them. However, cautious interpretation of the proposed mechanism is necessary. There is currently no experimental evidence on the dynamics of the Na+ and K+ currents at the mechanoreceptor level to support or disprove this view.
Experimental intracellular recordings from the soma of the primary neurons of the guinea-pig ileum enteric nervous plexus demonstrate that after application of 5-HT (Bertrand et al., 2000) the neuron produces long lasting, 2 - 4 s, trains of action potentials of amplitude 75-80 mV and a frequency ranging from 0.1 to 10 (Hz). Numerical analysis of the dynamics of the ion currents helps suggest the mechanisms responsible for the observed response. Thus, major changes are registered in the IpNa and IpK currents with high-frequency fluctuations recorded during the recovery phase. It is possible to assume that the increased excitability of the mechanoreceptors and instability of the Na+ and K+ currents decrease the threshold of activation of the AH neuron.
Selective activation of the 5-HT3 receptors on the soma of the S neuron of the guinea-pig ileum induces action potentials of 5 mV and of low frequency (Bertrand et al., 2000). These experimental findings are of particular interest because the physiological saline in the experimental preparation contained nicardipine and scopolamine - drugs that affect cholinergic transmission. The numerical simulation of co-transmission by ACh and 5-HT shows that the motor neuron generates high amplitude,134 mV and frequency,7.8 Hz, action potentials. This response can be attributed to a heteromeric co-assembly of the 5-HT3 receptors with the nicotinic ACh (α4 subunit) receptors (Van Hooft et al., 1998; Barajas-Lopez et al., 2001; Holler et al., 1999; Pindon et al., 2002). While these experimental facts concur with the results of our numerical simulations the putative role of the 5-HT3/nACh ligand-channel co-assembly requires further pharmacological investigation.
We were able to analyze numerically the effects of co-expression of the 5-HT3, 5-HT4 and nΑCh receptors on the soma of the S neuron on the dynamics of the electrical signal transduction. Our results demonstrate the minor role played by 5-HT type 3, in contrast to the 5-HT type 4 receptors. Activation of 5-HT4 receptors evoked a long-lasting train of fast action potentials. Co-stimulation of the (5-HT3 - nACh) and (5-HT4 - nACh) receptors has a considerable excitatory effect on the neuron. It resulted in a significant increase in the amplitude and frequency of firing rate. These findings are in line with the belief of possible co-assembly of serotonergic and cholinergic receptors (Legay et al., 1984; Briejer and Schuurkers, 1996; Foxx-Orenstein et al., 1998).
CONCLUSION
Comprehensive experimental data on the physiology and neuro-pharmacology of the myenteric nervous plexus, combined with modern computational modeling technology, have allowed us to study in a “virtual environment” the effects of co-transmission by 5-HT and ACh and of receptor polymodality, i.e., 5-HT3, 5-HT4, nACh and μACh type receptors, on its electrical activity. The numerical investigations helped us reveal intrinsic mechanisms of co-transmission at the cellular and tissue levels that could not have been elucidated using the existing experimental in vivo or in vitro methods.
REFERENCES
- Bertrand, P.P., W.A.A. Kunze, J.B. Furness and J.C. Borstein, 2000. The terminals of myenteric intrinsic primary afferent neurons of the guinea-pig ileum are excited by 5-hydroxytryptamine acting at 5-hydroxytryptamine - 3 receptors. Neuroscience, 101: 459-469.
PubMed








