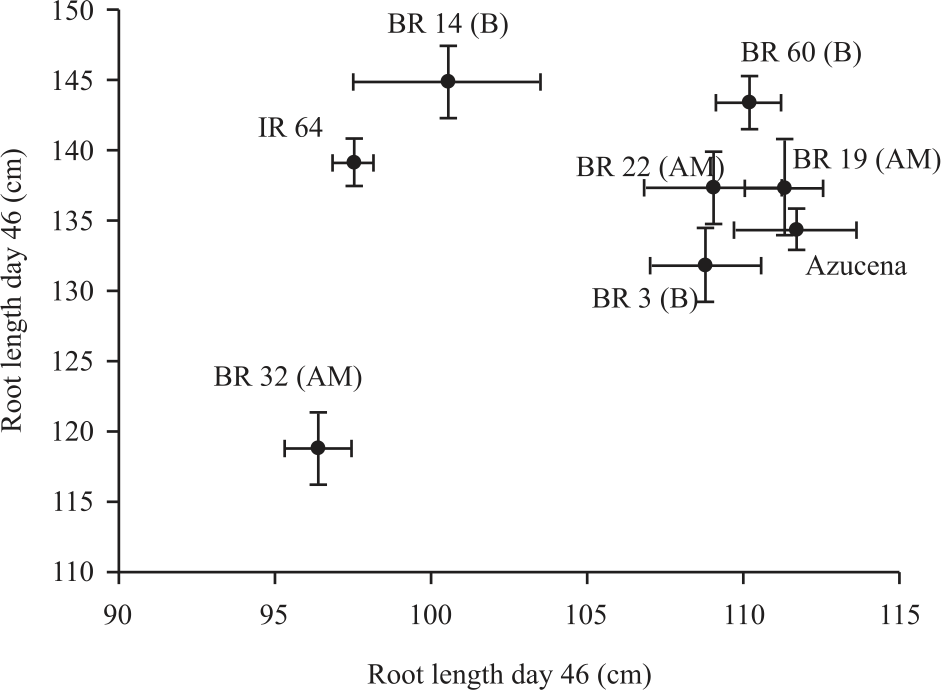
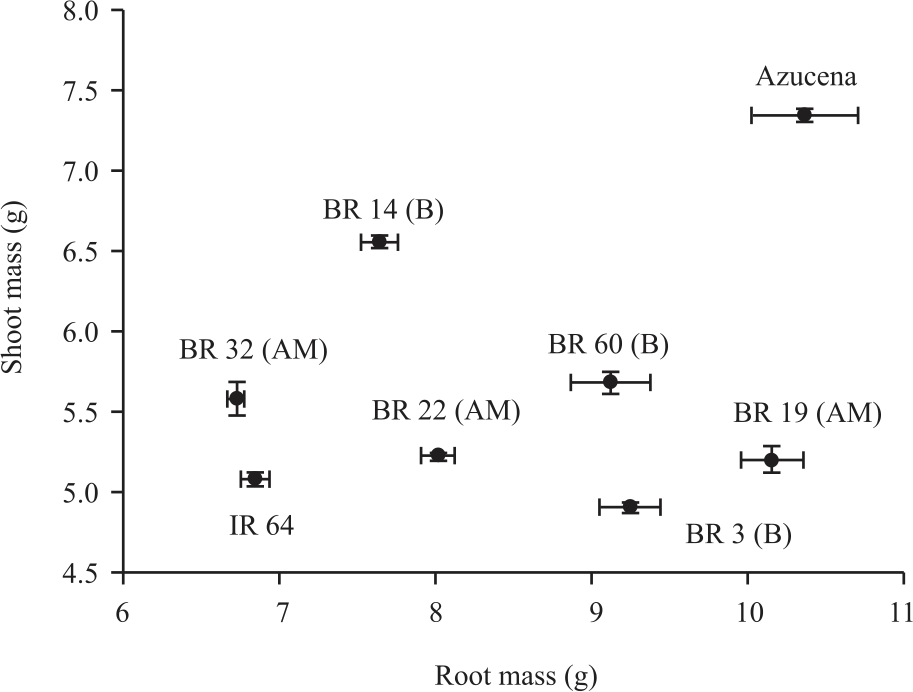
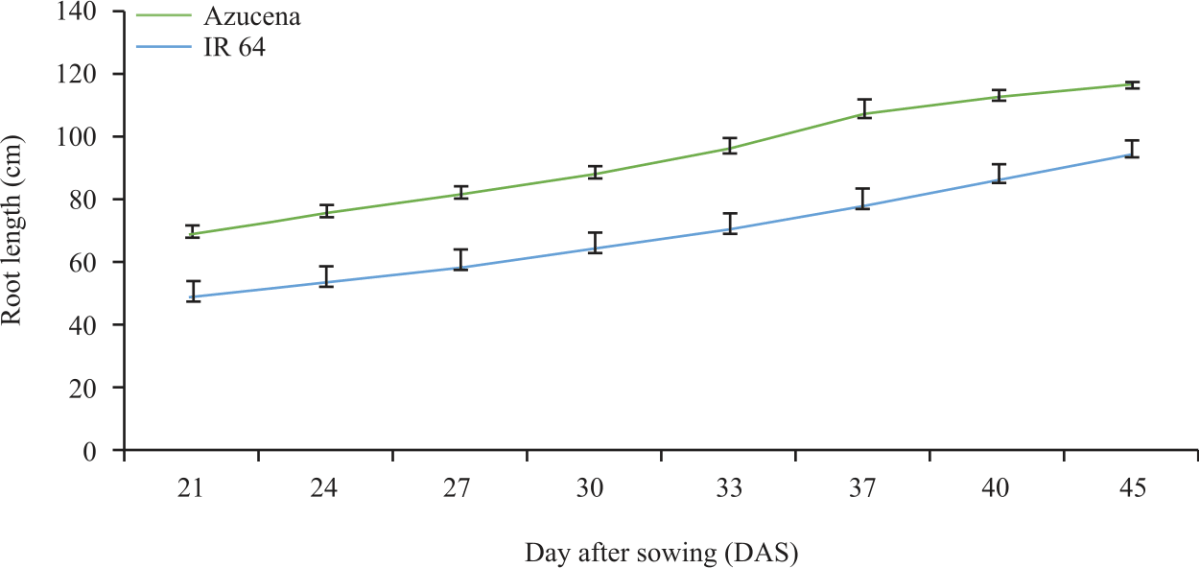
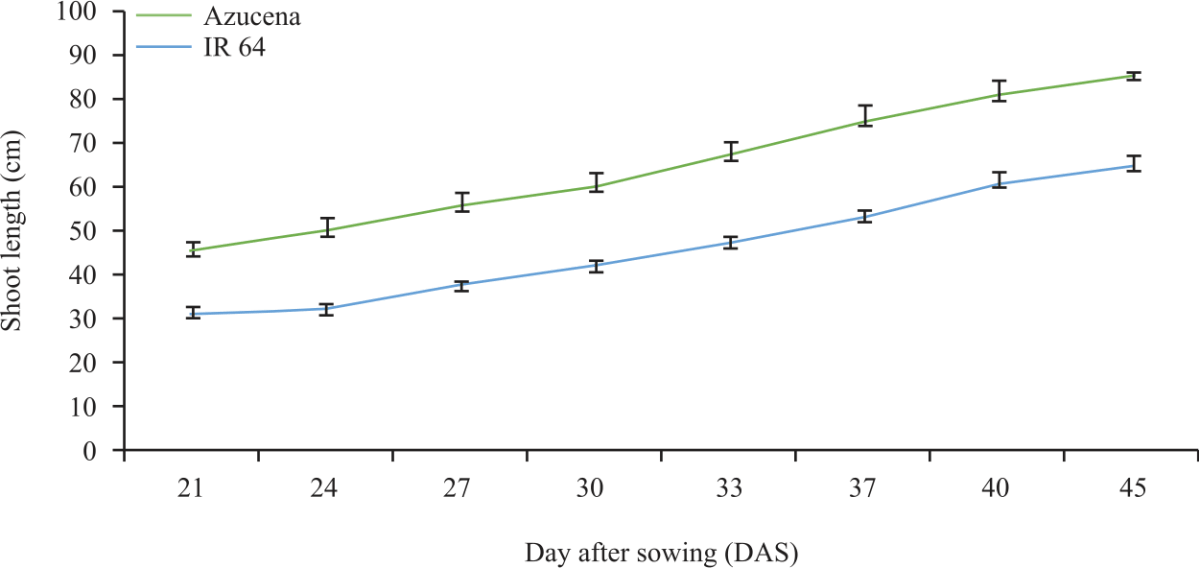
Research Article
Assessing Root System Characteristics in a Subset of Rice Accessions Through Hydroponic Rhizotron Systems
Department of Biochemistry and Molecular Biology, University of Chittagong, Chittagong-4331, Bangladesh
LiveDNA: 880.35589
Md Saiful Islam
Department of Biochemistry and Molecular Biology, University of Chittagong, Chittagong-4331, Bangladesh