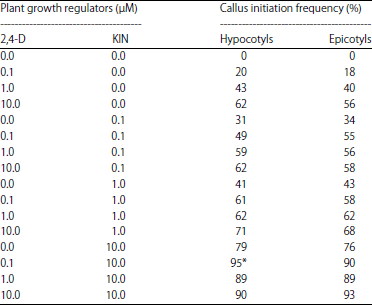
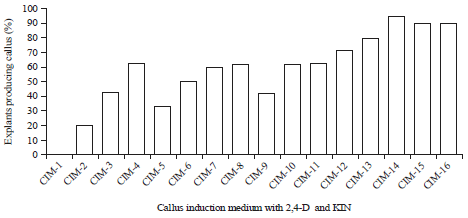
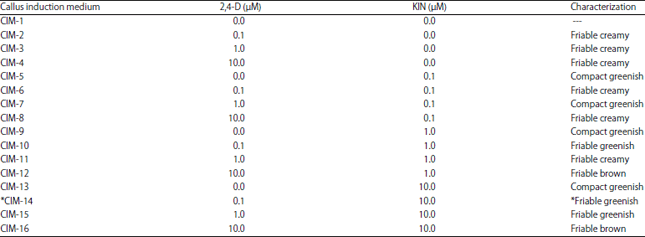
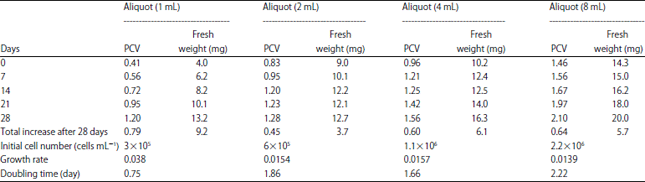
Research Article
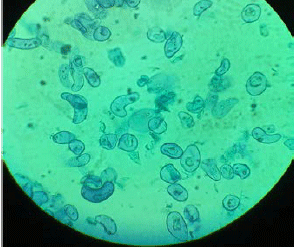
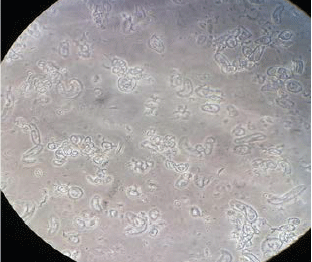
Callus Induction and Establishment of Cell Suspension Culture of Cumin (Cuminum cyminum L.)
Department of Biotechnology, P.S. Science and H.D. Patel arts College, Kadi-382517, KSV University, Gandhinagar, Gujarat, India
LiveDNA: 91.3464
P.N. Bhatt
Sun Agrigenetics Pvt. Ltd, Vadodara, India
D.P. Bhatt
Sun Agrigenetics Pvt. Ltd, Vadodara, India