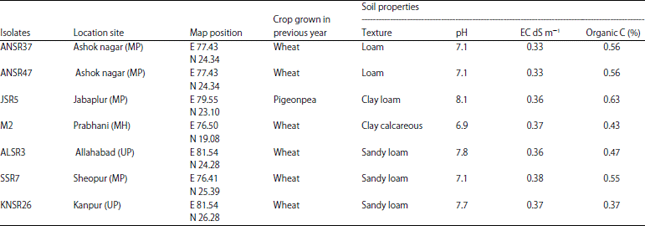
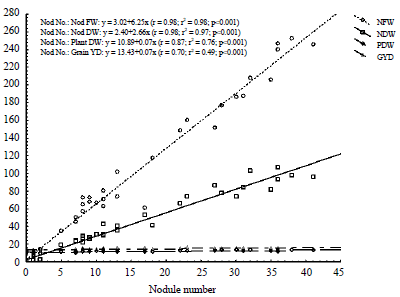
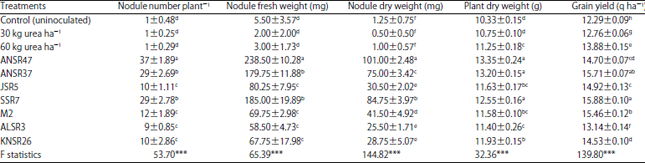
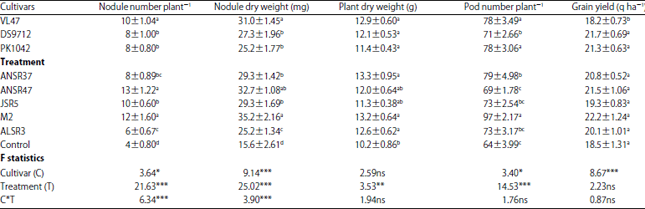
Research Article
Response of Soybean (Glycine max L. Merr) Cultivars to Soil Microsymbionts Inoculation in Agriculture Field in India
Microbiology Laboratory, Department of Genetics and Plant Breeding, Institute of Agricultural Sciences, Varanasi, India
LiveDNA: 91.3371
Akhil Anand
Microbiology Laboratory, Department of Genetics and Plant Breeding, Institute of Agricultural Sciences, Varanasi, India
Akhouri Vaishampayan
Microbiology Laboratory, Department of Genetics and Plant Breeding, Institute of Agricultural Sciences, Varanasi, India