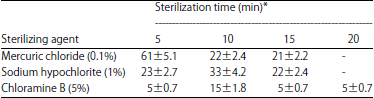
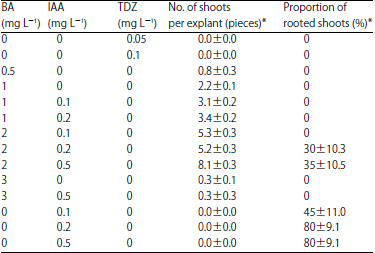
Research Article
A Method for Microclonal Propagation of Staurogyne repens in Tissue Culture
Southern Federal University, Rostov-on-Don, Russia
E.V. Lutsenko
Southern Federal University, Rostov-on-Don, Russia
V.A. Chokheli
Southern Federal University, Rostov-on-Don, Russia
LiveDNA: 712.13405
A.V. Vereschagina
Southern Federal University, Rostov-on-Don, Russia
K.Y. Rachkovskaya
Southern Federal University, Rostov-on-Don, Russia
V.S. Lysenko
Southern Federal University, Rostov-on-Don, Russia
LiveDNA: 712.13410
T.V. Varduny
Southern Federal University, Rostov-on-Don, Russia