Research Article
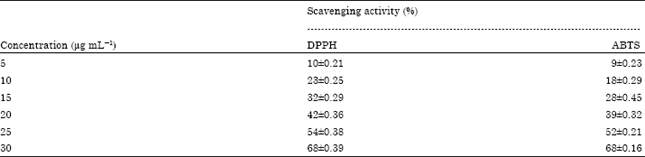
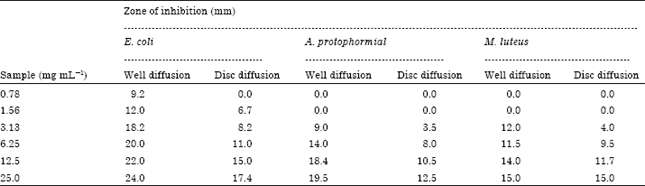
Antioxidant and Antibacterial Activity of Hippophae rhamnoides Methanolic Leaf Extracts from Dry Temperate Agro-climatic Region of Himachal Pradesh
Department of Pharmaceutical Chemistry, Rayat Institute of Pharmacy S.B.S Nagar Railmajra, Punjab, India
R. Sharma
Department of Pharmaceutical Chemistry, Rayat Institute of Pharmacy S.B.S Nagar Railmajra, Punjab, India
R. Arora
Department of Pharmaceutical Chemistry, Rayat Institute of Pharmacy S.B.S Nagar Railmajra, Punjab, India
M. Bali
Department of Pharmaceutical Chemistry, Rayat and Bahara College of Engineering and Biotechnology for Women, Mohali, India