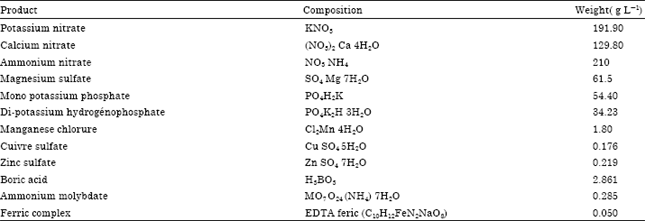
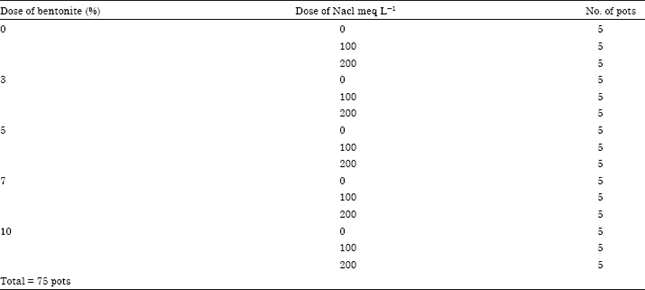
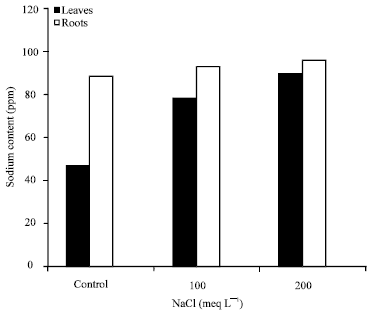
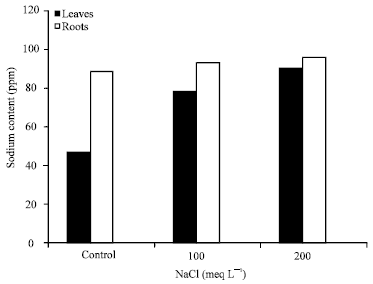
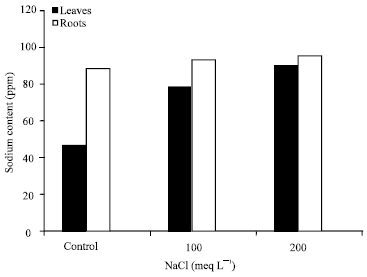
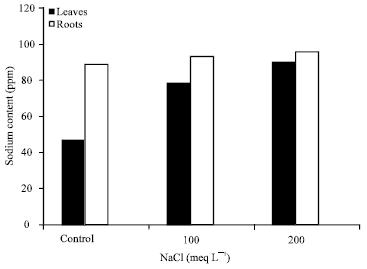
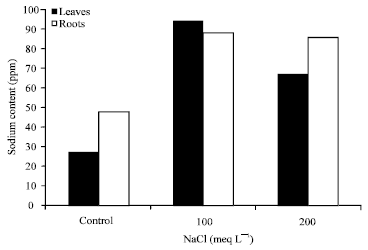
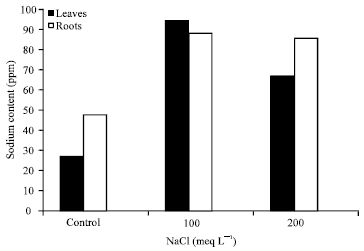
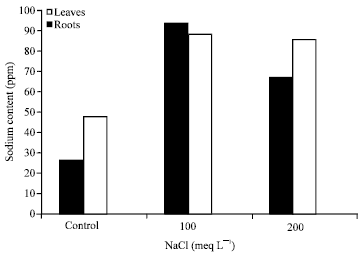
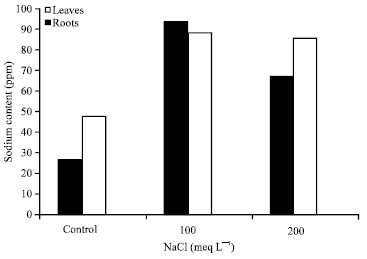
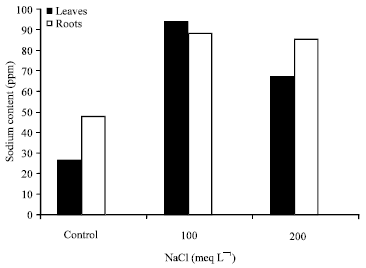
Research Article
Effect of Salinity and Bentonite on Mineral Soil Characteristics: Behavior Study of Leguminous Plant (Vicia faba L.)
Laboratoire de Biologie Faculte des Sciences, Universite de Mostaganem, route de Belhacel BP 923 27000, Mostaganem Algerie
A. Chibani
Laboratoire de Biologie Faculte des Sciences, Universite de Mostaganem, route de Belhacel BP 923 27000, Mostaganem Algerie
A. Bachir Bouyadjra
Laboratoire de Physiologie Vegetale, Faculte des Sciences, Universite d`Oran, Senia 31 000 Oran, Algerie