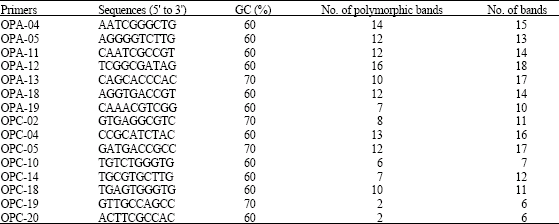
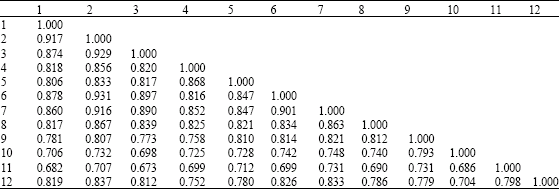
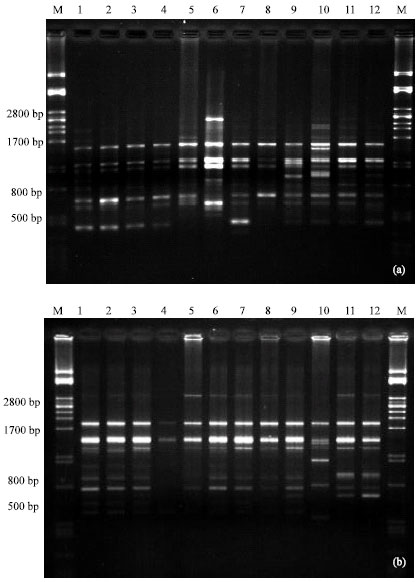
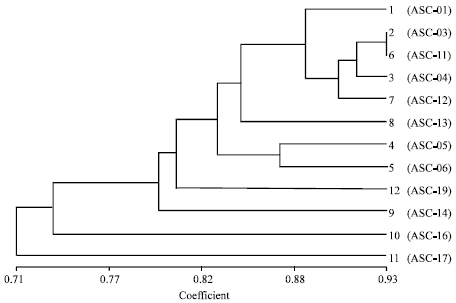
Research Article
Phylogenetic Analysis of Cucumis sativus Using RAPD Molecular Markers
Prapassorn D. Eungwanichayapant and Ekachai Chukeatirote School of Science, Mae Fah Luang University, Chiang Rai 57100, Thailand
Nittaya Laosat
Prapassorn D. Eungwanichayapant and Ekachai Chukeatirote School of Science, Mae Fah Luang University, Chiang Rai 57100, Thailand
Wannipa Suksawat
Prapassorn D. Eungwanichayapant and Ekachai Chukeatirote School of Science, Mae Fah Luang University, Chiang Rai 57100, Thailand
Siam Popluechai
Prapassorn D. Eungwanichayapant and Ekachai Chukeatirote School of Science, Mae Fah Luang University, Chiang Rai 57100, Thailand
Prapassorn D. Eungwanichayapant
Prapassorn D. Eungwanichayapant and Ekachai Chukeatirote School of Science, Mae Fah Luang University, Chiang Rai 57100, Thailand
Ekachai Chukeatirote
Prapassorn D. Eungwanichayapant and Ekachai Chukeatirote School of Science, Mae Fah Luang University, Chiang Rai 57100, Thailand