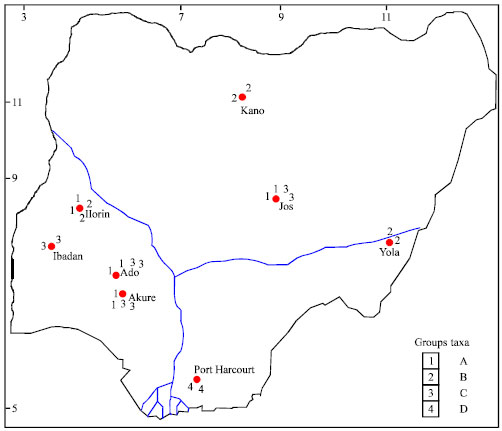
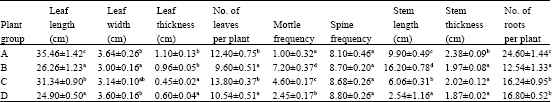
Research Article
Comparative Study of Vegetative Morphology and the Existing Taxonomic Status of Aloe vera L.
Department of Soil Science and Technology, Federal University of Technology, P.M.B. 1526 Owerri, Nigeria
A. C. Odiyi
Department of Soil Science and Technology, Federal University of Technology, P.M.B. 1526 Owerri, Nigeria