Research Article
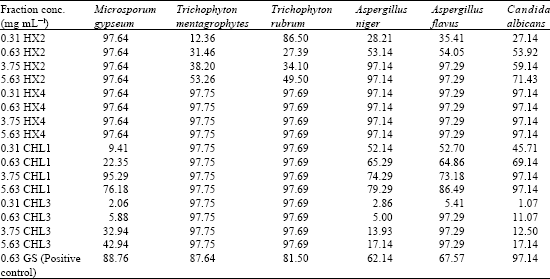
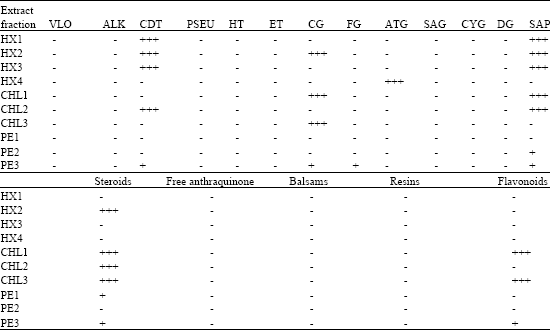
Antifungal Activity and Phytochemical Analysis of Column Chromatographic Fractions of Stem Bark Extracts of Ficus sycomorus L. (Moraceae)
Department of Biochemistry, Usmanu Danfodiyo University, P.M.B. 2346, Sokoto, Nigeria
M. Lawal
Department of Biochemistry, Usmanu Danfodiyo University, P.M.B. 2346, Sokoto, Nigeria
B. Y. Muhammad
Department of Pharmacology, Usmanu Danfodiyo University, P.M.B. 2346, Sokoto, Nigeria
R. A. Umar
Department of Biochemistry, Usmanu Danfodiyo University, P.M.B. 2346, Sokoto, Nigeria
L. S. Bilbis
Department of Biochemistry, Usmanu Danfodiyo University, P.M.B. 2346, Sokoto, Nigeria
U. Z. Faruk
Department of Pure and Applied Chemistry, Usmanu Danfodiyo University, P.M.B. 2346, Sokoto, Nigeria
A. A. Ebbo
Department of Veterinary Physiology and Pharmacology, Usmanu Danfodiyo University, P.M.B. 2346, Sokoto, Nigeria