Research Article
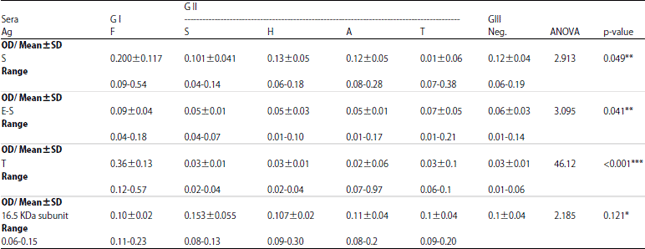
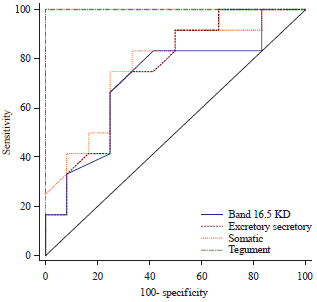
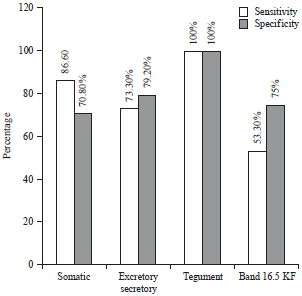
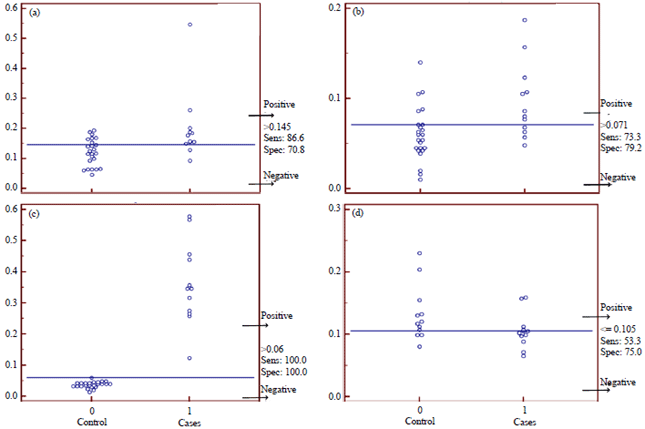
Comparative Serodiagnostic Evaluation of Tegumental Antigen and its 16.5 KDa Subunit to Crude Antigens in Human Fascioliasis
Faculty of Medicine, Ain Shams University, Cairo, Egypt
R.M. Sarhan
Faculty of Medicine, Ain Shams University, Cairo, Egypt
LiveDNA: 20.24817
A.F. Badawy
Faculty of Medicine, Ain Shams University, Cairo, Egypt
G.H. Shinkar
Faculty of Medicine, Ain Shams University, Cairo, Egypt